Pyrogallol is one of those chemicals that traces its roots well before the modern chemical industry. Born from a period when plant extracts served as both medicine and foundation for experimental chemistry, its discovery is usually credited to the German chemist Carl Wilhelm Scheele in 1786. He uncovered the substance while studying gallic acid—himself part of an era obsessed with parsing nature’s secrets from oak galls and similar materials. Throughout the nineteenth century, pyrogallol found much of its early use in the realm of photographic development. Early black-and-white photography transformed once scientists realized that this modest crystalline powder could reduce silver salts to metallic silver, delivering sharper images and more manageable developing times. Later, it carved out its place in analytical labs, becoming critical for oxygen absorption experiments. Without this step, understanding processes like respiratory metabolism or industrial gas mixtures would have been slowed down by years. These foundations matter today; we stand on decades of trial and creative repurposing that keep informing industrial and research applications even now.
Pyrogallol matters today for more than its legacy. Its formula—C6H3(OH)3—makes it a simple yet uniquely reactive compound. Laboratories turn to it for specialized oxidation and reduction work. Water treatment plants and gas analysis setups still count on its knack for soaking up oxygen efficiently. Its presence in dye manufacturing, hair coloring, and even in certain clinical diagnostics keeps the compound relevant. While other more stable or less toxic chemicals have nudged into its traditional roles, pyrogallol’s combination of reactivity and reliability keeps it in regular rotation.
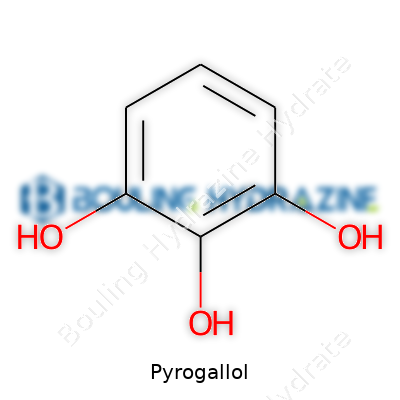
Appearance often gives a first clue with chemicals—pyrogallol’s white crystalline powder seems unremarkable but hides plenty of bite. Hydrated or exposed to air, the substance slowly darkens as it oxidizes. It dissolves well in water and alcohols, giving it flexibility for varied tasks. Because of its three adjacent hydroxyl groups on a benzene ring, the reactivity goes much higher than with more familiar single-variant phenols. This high reactivity brings both utility and challenge; the properties that allow rapid oxygen absorption also create hazardous exposure scenarios if not handled with care. Those dark stains on fingers or glassware in the lab? That comes from the compound’s sensitivity to air. It’s one of those small cues that remind chemists of what they’re handling long before the toxicological data comes into play.
Commercial pyrogallol comes marked as high-purity or technical-grade depending on its target use—analytical labs demand the purest stock, while dye manufacturers sometimes work with looser standards. Labeling requirements follow regional chemical safety rules, calling out the substance’s tendency to oxidize and its toxicity profile. The demand for clear specificity in labeling and documentation springs not from bureaucratic habit but the lived reality of dealing with sensitive, potentially dangerous reagents. Mislabeling or casual handling has triggered a long string of toxic exposures, teaching the chemical community the hard way about respect for detail.
Most manufacturers produce pyrogallol by heating gallic acid—a process called dry distillation. In less-glamorous terms, workers heat the gallic acid in a closed vessel, allowing the pyrogallol to distill over, followed by cooling and purification. This method holds up because of its straightforwardness; it doesn’t require exotic reagents or advanced technology. At scale, tweaks in temperature, pressure, and purification have shaved costs and increased safety. As green chemistry principles grow in influence, researchers keep looking for cleaner and less energy-hungry production methods, but for now, tradition persists for reasons of both cost and process control.
Pyrogallol sets a brisk pace once it enters the mix. Its three closely spaced hydroxyl groups not only boost its ability to donate electrons but also unlock a menu of follow-on reactions—oxidation with atmospheric oxygen stands out as nearly instant, forming quinones and colored byproducts. Analytical chemists love this trait, using pyrogallol solutions as quick oxygen indicators by gauging the color change. It can also undergo methylation or acetylation to build derivatives used in specialty dye industries. For those thinking outside the box, researchers tune these core reactions to develop new sensing agents and polymerization catalysts. Exploring the space where pyrogallol meets other energetically-inclined chemicals continues to drive work on medical diagnostics and advanced material science.
Those who spend time in the chemical trade or at the research bench know pyrogallol answers to multiple names. Common synonyms include 1,2,3-trihydroxybenzene, pyrogallic acid, and benzene-1,2,3-triol. Older literature sometimes tags it under photogen or oxidol, especially where photographic chemistry enters the conversation. Different suppliers promote brand-specific versions, but the substance itself goes under these core names throughout most scientific literature and regulatory catalogs.
Any review of pyrogallol means getting real about safety. The stuff stains not just fingers but lungs and livers if exposure goes unchecked. Strict workplace ventilation rules, personal protective equipment, and airtight storage aren’t open to skimping here. Medical case studies and regulatory guidelines both point out acute poisoning risks—including nausea, confusion, and harm to blood cells. Workers need clear training, quick cleanup protocols, and access to medical guidance. In laboratories, even short-term exposure gets flagged as a risk, thanks to its ability to absorb rapidly through skin and mucous membranes. Waste disposal complicates things as well, since pyrogallic solutions contaminated with heavy metals or other reactants build up dangerous residues that require secure management. Operational standards grow tighter as experience highlights where old habits fall short, making a strong case for behavioral and systemic safety culture.
Pyrogallol still finds its way into water treatment, hair dye manufacture, and analytical laboratories. The oxygen absorption trick keeps it critical in controlled-atmosphere experiments, gas purity testing, and metabolic measurements. These uses often fly beneath the public radar, but plenty of research scientists and process engineers keep the material on hand for workhorse chemical roles that resist easy substitution. While hair dye applications draw scrutiny due to toxicity risks, the compound’s advantages in color-fastness and pigment development keep it in the product mix for certain specialty shades. The dye industry counts on the deep, durable tones it delivers, even as regulatory pressure grows. Silver-based photography, though now largely historical, owes much of its early efficiency and image quality to pyrogallol-based developers. Researchers in the environmental sciences also employ it for oxygen-trapping studies, supporting climate modeling and industrial hygiene surveys.
Looking at current research, pyrogallol stands out for the versatility built into its basic structure. Synthetic chemists keep reaching for its triol framework to build sensors aimed at detecting hydrogen peroxide or other reactive oxygen species. These applications show up in medical diagnostics and environmental monitors. As computational chemistry matures, teams work to predict new derivatives and reaction pathways that could improve treatment of neurodegenerative diseases. The molecule forms a springboard for exploring antioxidant and pro-oxidant mechanisms in biochemistry, often shedding light on aging, oxidative stress, and underlying cellular damage in disease processes. Pyrogallol’s combination of strong reactivity, available derivatives, and defined toxicity forces R&D teams to balance innovation against practical safety and regulatory navigation. Trends show a push for safer analogues that retain the compound’s best traits but with lower toxicity—reflecting how old workhorse chemicals still fuel new science, provided the risk is managed intelligently.
Decades of studies link pyrogallol to a known pattern of toxic responses in lab animals and accidental human exposures alike. Absorbed through skin or inhaled, it can damage red blood cells, induce anemia, and trigger significant organ toxicity at relatively modest doses. Even though acute poisonings aren’t commonplace in well-managed labs or industries, accidental spills and low-level chronic exposures have left a long enough trail in published literature to provoke caution. Regulatory agencies place a hard spotlight on its carcinogenic and mutagenic potential, leading to phased withdrawal from consumer-facing products whenever safer substitutes come into play. Some of the most telling research concerns trace environmental contamination, where improperly treated waste delivered downstream has occasionally triggered ecosystem damage—small but hard lessons in treating every reactive compound with respect. Reviewing the evidence, both old-timers and newcomers to the field see clearly that efficiency never justifies underplaying toxic risk. Building a culture that prizes monitoring, transparent reporting, and ongoing review of usage conditions pays off in both safety and science.
Chemistry’s classics either fade away or evolve with the times. Pyrogallol lands somewhere in the middle; its signature strength in redox reactions and dye development keeps it in play, but industrial users and researchers both realize its limitations, especially where health and environmental impacts outshine raw chemical performance. Interest keeps shifting toward derivatives designed for lower toxicity and new analytical purposes. Meanwhile, green chemistry principles point R&D investments toward alternative synthesis routes with less energy, lower emissions, and reduced byproduct creation. The story here involves choices: keep using a proven tool with acknowledged hazards, or invest in creating safer, equally reliable substitutes. Demand for sharp, selective sensors and new medical diagnostics hints at a future where pyrogallol’s legacy continues in modified forms built for tighter regulations and improved safety. The overall message? Progress depends on acknowledging what made pyrogallol so useful and where its risks now demand a better approach.
Pyrogallol rarely gets much attention outside labs and textbooks, but it quietly serves a few important niches, especially in chemistry and industry. My time tripping over beakers in a university lab gave me a close look at the stuff, and I remember the stained hands and the snappy smell. This isn't something you see in your home kit, but understanding its story explains a lot about how old-school science shapes industries—even today.
The days of standing in a darkroom with developer trays are almost gone, replaced by pixel-perfect screens and one-click filters. Even so, those who still love black-and-white photography might know pyrogallol as one of the secret ingredients for certain film developers. Classic formulas, like those used by Ansel Adams, worked their magic partly thanks to this white, crystalline powder. The chemical offers fine grain and a certain sharpness to film, making it prized by serious photographers who cling to analog methods. Film depends on carefully controlled chemical reactions, and pyrogallol’s predictability made it a favorite before digital photography shifted the whole landscape.
Ask any chemist about testing for oxygen, and pyrogallol probably pops up. Drop it in a solution, add some oxygen, and you’ll see a dark brown color shoot through—nature’s own indicator. This simple test harnesses pyrogallol’s tendency to oxidize rapidly. It never failed to impress at science fairs, but its real value shows in more than just classrooms. Laboratories once relied on this reaction to measure oxygen content, especially in gas analysis. Technology gave us new sensors and digital readers, but smaller labs, or those working in resource-limited settings, sometimes still use the old pyrogallol test because it gets the job done with little fuss.
One place pyrogallol keeps cropping up is dermatology. Doctors have used it to treat conditions like psoriasis, taking advantage of its ability to destroy cells and slow abnormal growth on the skin. Modern medicine pushed pyrogallol aside in favor of newer therapies with fewer risks—the substance can be toxic and irritate skin or be absorbed into the body if mishandled. It reminds us that progress in health means safer tools, but also that older methods sometimes hang on in places where new options seem out of reach. This raises the stakes for making affordable, safer alternatives widely available so no one ends up stuck with riskier treatments.
Industries still draw on pyrogallol for creating dyes and inks, especially those that demand a specific shade or property. Leatherworkers, textile makers, and people involved in specialty printing know its value. At the same time, handling pyrogallol carries dangers—both acute exposure and long-term contact bring health concerns. Strict workplace safety and proper training keep workers safe, but accidents and shortcuts happen. Rather than blame the chemical, the focus should stay on replacing it where possible or strengthening safety culture where it remains essential. If better, less-toxic options win out in the end, everyone, including the environment, comes out ahead.
Learning from pyrogallol’s story pushes us toward more thoughtful choices in science and industry. Holding onto the good while discarding the harmful parts depends on research, open conversation, and investment. Pyrogallol may never return to the center of the spotlight, but approaching all chemicals with the same respect and caution helps keep progress on the right path.
Pyrogallol often doesn’t get much attention outside the circles of chemists and lab workers. Still, its strong role in certain industries—like photography, hair dyes, and chemical research—calls for real know-how about safety. Speaking from ten years spent working in a university lab, too many assume basic gloves and a lab coat will cut it. Pyrogallol tells another story. Just one whiff too close, or a careless moment with bare skin, brings home the urgency of smart handling.
A lot of chemicals trigger warnings, but a few like pyrogallol back them up with quick, hard-hitting effects. It’s toxic if absorbed or inhaled and can irritate skin, eyes, and the respiratory system. It tends to stain anything it touches, too, leaving no doubt when an accident happens. Nobody forgets the first time a brown patch appears on their lab coat—or worse, on their fingers that tingle and itch for the rest of the week.
Headache, nausea, and dizziness can sneak up if pyrogallol slips through your defenses. On top of that, pyrogallol reacts with air and other chemicals, sometimes releasing harmful vapors. I’ve seen reactions get out of hand when open containers let fumes escape, especially in smaller or poorly ventilated labs.
Some rules might sound obvious, but daily practice turns advice into second nature. This starts with gloves—nitrile, not latex, since pyrogallol gets through some materials too easily. Lab coats and goggles come next. More than once, I’ve seen a splash miss someone’s face only because those goggles didn’t slip off. I keep my sleeves rolled down, which goes against the comfort of short sleeves but saves skin.
Ventilation deserves real respect. Working inside a fume hood isn’t just for show. I learned the hard way that even brief exposure to drift from an open container leads to sore throat by the end of the day. Good airflow, frequent breaks, and a closed container combine for noticeable comfort and safety.
No matter how careful you feel, spills can still happen. My go-to move is to have spill kits ready—sawdust or special absorbent powder keeps a minor slip from turning into a big problem. Immediate cleanup keeps residue from building up on benches and floors.
Washing hands thoroughly before leaving the workspace can’t be skipped. I’ve worked with colleagues who ended up with stubborn stains at home just from rushing out for lunch. Everyone using the space should keep this habit.
Nothing replaces training when it comes to handling chemicals like pyrogallol. Every new team member I work with gets a run-through on specific hazards and response steps. Clear labeling keeps everyone aware, even visitors unfamiliar with the setup. Errors sometimes come from people not knowing what’s in the bottle—labels cut down those surprises.
Health and safety culture grows from sharing stories and lessons, not just posting rules. As someone who’s been on the receiving end of a chemical mishap, I know stories stick in the mind in a way rules never do. That’s what keeps people careful and looking out for each other during hectic or long days.
Anyone who's spent time in a research lab quickly learns that Pyrogallol asks for more respect than many other fine chemicals on the shelf. This compound — used to make dyes, test for oxygen, and craft photographic developers — reacts quickly with air and moisture. It's not just a quirky trait. Exposing it to humidity or air leads to spoilage, even riskier situations if stored near heat. I remember the lesson well from a grad school lab, where we discovered brown stains creeping up an unsealed bottle — all because we forgot to screw the lid down and return it to the desiccator. That mess cost us a full afternoon and almost ruined the controls for our experiment.
Forget idealized laboratory pictures with bottles in open air. Pyrogallol does best in an airtight glass container, away from sunlight. Even my most organized chemistry professor rarely trusted it in plastic, since lingering moisture gets trapped too easily. Failing to keep the jar tightly sealed welcomes an ugly, sticky crust — not what you want seconds before making a photographic developer or running sensitive oxygen tests. My own lab built the habit of always double-checking caps and adding a clear “Desiccator Only” label on the bottle. No one wanted to risk a shelf full of clumped, degraded powder or the hassle of ordering emergency replacements mid-project.
Pyrogallol can ignite at higher temperatures. Leaving containers near a radiator or sunny windowsill isn’t just absent-minded; it’s dangerous. Reports of accidental fires may be rare, but mistakes stack up around old heating vents or cluttered benchtops. Good labeling and discipline keep the risks down. Including date-opened notes on bottles helps spot spoilage early. Fresh powder comes white, not brown. Regular checks aren’t just for safety audits. They save time and money, since spoiled material can botch experiments or slow industrial production. Many researchers keep smaller working stocks, refilling from a large, well-protected batch in the storeroom as needed.
Some chemicals only seem intimidating because no one explained simple safeguards. Pyrogallol rewards a basic approach: dry, cool, sealed, and away from high-traffic spaces. That’s how the bigger facilities avoid messy problems and how small university labs protect tight budgets. A little upfront care — tight cap, glass jar, desiccator or properly climate-controlled cabinet — prevents bigger headaches. My own workflow improved once I carved out a dedicated space for sensitive reagents, borrowing tips from professional suppliers who prioritize safety over convenience.
I’ve seen the fallout from careless storage, from ruined batches to impromptu cleanups. Pyrogallol’s quirks teach patience and attention to detail. If newcomers approach it with respect and institutions back them with solid training, most headaches can be avoided. Chemistry always demands vigilance, but that vigilance pays off in safety, savings, and fewer disruptions to discovery.
You’ll sometimes hear the name “pyrogallol” in passing within labs, in old photographic handbooks, or within books on dyeing. To answer the textbook question: its chemical formula is C6H6O3. Simple as that might look, those nine letters and numbers open the door to a compound that’s written itself into science and industry for generations.
This compound goes way beyond labels on brown glass bottles. Pyrogallol shows up in tests for oxygen—used by chemists who track air quality or measure dissolved oxygen in water. The reaction is eye-catching: it turns rapidly from white to brown as it reacts with oxygen, helping folks literally “see” the invisible gases moving around us. That property, rooted in its formula, gives pyrogallol an edge as a real-world problem-solver.
In the shifting landscape of photography, pyrogallol helped early photographers bring their images to life. Black-and-white prints developed with it show a punchy contrast and a different grain structure that some artists still chase today, despite safer or more convenient options being available now.
Walk through the history of dyes and inks, and pyrogallol shows up again. This molecule helped tint clothing, paper, and sometimes even hair. That speaks to its chemical flexibility, which comes straight from the structure of C6H6O3—a benzene ring loaded up with three hydroxyl groups, always ready to latch on to something new.
But the deeper story isn’t always positive. Repeated studies have flagged pyrogallol’s toxicity if handled carelessly. It can cause skin irritation, breathing difficulty, and even more serious health issues in high concentrations. The International Agency for Research on Cancer (IARC) has even looked closely at its risky side. That means anyone working with this compound, whether in classic photographic processing or niche chemistry, needs to respect those risks and follow sound lab practices.
There’s a lesson in every bottle of pyrogallol on a lab shelf. The right gloves, solid ventilation, and proper chemical waste management turn a risky element into a useful tool rather than a source of accidents or environmental mishaps. New researchers—especially those coming into chemistry or art restoration fields—can benefit from training not just in technique, but in respect for what each compound represents. Mistakes caused by ignorance or carelessness have pushed many labs and companies to substitute safer alternatives wherever possible, but the old methods—and old risks—sometimes stick around for the sake of quality or tradition.
Science keeps moving in the direction of safety. Green chemistry experts keep pushing the boundaries, hunting for organic or less toxic substitutes that can fill the gaps left where pyrogallol once ruled. But for certain historical processes, and rare analytical tests, there isn’t always a ready swap. In these cases, lab safety isn’t just best practice; it’s essential.
Whether viewed as a relic of 19th-century science or a modern specialty chemical, pyrogallol’s story reminds us how chemistry shapes the world. Understanding the formula C6H6O3 is more than trivia—it’s a doorway to learning how tools, risks, and choices connect in a real-world lab or workshop, and why treating every substance with thought and respect makes all the difference.
Plenty of folks working in research labs or old-school photography have come across pyrogallol, a chemical made famous for its role in developing black and white photos and testing metals. At first glance, a bottle sitting on a shelf might not seem like much of a risk. Reality says different. Reports and studies from credible health agencies, including the National Institute for Occupational Safety and Health (NIOSH), have raised genuine concerns about how pyrogallol affects human health. Having handled similar compounds in the past, I can say that a little respect for these chemicals goes a long way.
Pyrogallol comes with some baggage. It’s toxic—through skin contact, inhalation, and if it’s swallowed. Symptoms from exposure go beyond a quick headache. People exposed to dust or vapor can experience irritation of the skin, eyes, and respiratory tract. Mild irritation is often just the start. In heavier doses or prolonged contact, things get dangerous. Accidental spills or careless handling have led to severe burns, tissue necrosis, and in rare cases, systemic toxicity.
The chemical works by stealing oxygen from your blood, something even seasoned lab techs fear. When oxygen levels drop in the body, the results can roll out quickly: dizziness, confusion, even collapse. Some studies have found repeated or large exposures damage internal organs, including the liver and kidneys.
While animal testing doesn’t always translate directly to humans, repeated research from toxicology labs shows that pyrogallol can cause mutations in cells. Long-term risks remain unclear, but enough evidence hints at carcinogenic potential for all but the boldest risk-takers to take pause. The United States Environmental Protection Agency and major European safety boards flag pyrogallol for its acute toxicity and the potential for more chronic issues.
Beauty products once relied on it for hair dyes, but regulators stepped in. Most countries now ban or limit its use in cosmetics because of the threat to both clients and stylists. That’s a real-world example of evolving knowledge shaping societal choices. Smart policy follows solid science.
Memories of undergrad chemistry remind me how easy it is to underestimate substances that sound almost quaint. Proper gloves, mask, and eye protection felt like overkill at first, but tales floating around lab break rooms kept everyone vigilant. One slip with a toxic chemical makes an impression you don’t forget—sometimes literally.
Fact is, pyrogallol doesn’t belong in everyday settings. Laboratories still use it, but only behind fume hoods and with strict protocols in place. Even then, experienced workers keep spill kits nearby. Regular training, real accountability, and updated hazard labels all factor into responsible use.
Long-term, safer substitutes—notably for photographic or industrial applications—look more appealing. Some industries have phased out pyrogallol in favor of modern alternatives, reducing the number of hands that ever touch the stuff. Schools and hobbyists can pick less hazardous chemicals without sacrificing results. Safety data and open discussion help keep these improvements moving in the right direction.
Everyone from researchers to small-batch artisans benefits from knowing what they’re working with. Transparent warnings, fully funded regulatory oversight, and improvements in chemical education limit accidental exposures. Plain talk about risk—rooted in real evidence—sets the right tone for health and safety.