Talking about Mesitaldehyde brings up a mix of tradition and innovation wrapped up in a single chemical. Chemists going back a hundred years or so have gravitated toward the aromatic aldehydes, and Mesitaldehyde found its place not by accident. It slipped into the spotlight thanks to the fascination with methylated benzenes in the late 1800s, where scientists pushed further to see how they could tweak basic aromatic rings. Mesitaldehyde, with its three methyl groups giving it a strong presence, grew out of the curiosity baked into organic synthesis labs of that era. It’s easy to see that its introduction connected with the push for improved intermediates in dye and pharmaceutical production—a running theme in chemistry since coal tar days.
Mesitaldehyde pops up as a colorless or pale yellow oily liquid, depending on purity and handling. A strong, aromatic scent sets it apart from plainer compounds, and anyone who’s spent time in a synthetic lab will recognize its pungent presence. Business-wise, it’s found mostly as a laboratory intermediate, but niche industrial processes use it thanks to its bulky aromatic backbone. Specialty fragrances, custom polymers, and advanced pharmaceutical scaffolds anchor its small yet steady demand.
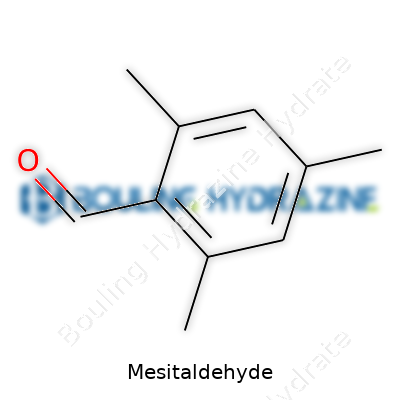
For those who’ve handled it, Mesitaldehyde’s slippery, low-volatility texture stands out. The molecular formula is C9H10O—implying a solid core of three methyls and a formyl group firmly attached to a benzene ring. This blocks out much of the reactivity seen in simpler aromatic aldehydes and adds bulk. Anyone working with it knows its melting point sits pretty low, so you’re mostly dealing with a liquid at room temperature. Solubility in common organic solvents like ether, chloroform, and ethanol makes it a go-to for synthesis, yet it avoids water, mostly floating atop any aqueous layer. Chemically, the methyl groups block harsh oxidation and reduce the rate of many routine electrophilic substitutions, nudging chemists to use stronger or more selective reagents for transformations.
Packed and labeled under various purity grades, Mesitaldehyde’s bottles in a lab sport names like “2,4,6-Trimethylbenzaldehyde” and a set of hazard tags. Standard purity hovers above 95%, but research work sometimes calls for extra care with distillation or chromatography to banish trace byproducts. Regulatory bodies flag it as an irritant, and you’ll spot flammable symbols on every shipment.
Synthesizing Mesitaldehyde can get a bit technical. The starting block, mesitylene (1,3,5-trimethylbenzene), meets a formylation agent, most commonly through an adapted Reimer–Tiemann or Gattermann–Koch protocol. Labs switching to Vilsmeier–Haack chemistry find tighter control, though results vary with scale, moisture, and operator skill. Purification follows with vacuum distillation to knock out byproducts and stabilize the product. Even with modern methods, the yield swings depending on reagent balance and temperature control—something any synthetic chemist learns after a few runs.
Mesitaldehyde resists the kind of aggressive chemical changes that work well on less-protected aromatics. Its methyl groups serve as shields in many ways. The aldehyde can participate in condensation reactions—think about aldol or Knoevenagel types—though steric hindrance sometimes reduces yields. Oxidizing it leads to 2,4,6-trimethylbenzoic acid, but you need tough oxidants. The bulky ring sees use as a starting point for more complex molecules in pharmaceuticals, specialty dyes, and even polymer additives, all because the core resists unwanted rearrangement or decomposition.
Mesitaldehyde goes by 2,4,6-Trimethylbenzaldehyde in chemical catalogs, with older literature sometimes referencing it as mesityl aldehyde. Anyone sourcing it will notice both names used interchangeably in patent filings and synthesis guides.
Safety practices around Mesitaldehyde reflect experience more than theory. Its volatility is low, but skin and eye irritation are sure bets if you let it spill or splash. Fume hoods, gloves, and good ventilation—not just checkboxes on a safety form, but real barriers against problems. Spills need adsorption and scrupulous floor cleaning to avoid stubborn odors. Despite its modest flammability, open flames don’t mix well with any aromatic aldehyde—too easy to lose a batch, or worse, touch off a fire. Emergency plans matter far more than the likelihood of an accident suggests. Seasoned chemists pass on this culture of caution.
Mesitaldehyde isn’t a bulk commodity chemical, so its fans sit in specialty labs and small-scale plants. It slips smoothly into varied reactions that build up complex organic skeletons—especially where crowding on the aromatic ring blocks unwanted side reactions. Natural product synthesis, specialty fragrance and flavor design, and pharmaceutical intermediate production suck up most of the world’s output. Additive manufacturing and polymer chemistry sometimes take an interest, relying on Mesitaldehyde’s rigid structure to impart stability and tweak physical properties of new materials.
Academic papers and industrial patents alike point to Mesitaldehyde’s unique role in pushing the boundaries of aromatic chemistry. Synthetic routes evolve as researchers adapt greener catalysts and try to boost yields, while minimizing noxious byproducts. Some labs explore biocatalysis—using engineered enzymes to formylate mesitylene in living cells—as a cleaner approach. Others chase down new reactions triggered by the combination of bulky methyls and a reactive aldehyde group, hoping for novel pharmaceuticals or advanced composite materials. My own digging into research articles suggested genuine excitement about pathways that combine Mesitaldehyde with newer catalytic systems, which might someday make this chemical more accessible for high-volume applications.
Safety data for Mesitaldehyde remains a constant priority, and for good reason. Standard toxicity screenings suggest it doesn’t behave much differently from similar aromatic aldehydes—irritant, potentially harmful if ingested, acutely toxic in large doses. Chronic exposure research barely scratches the surface, so cautious handling is the only responsible answer. Reports of environmental persistence pop up from time to time, usually tied to improper disposal or wastewater contamination. That risk matters, since aromatic compounds often resist natural breakdown, so green disposal and process control aren’t just empty gestures.
Looking at what’s next, Mesitaldehyde’s role probably stays in specialty applications, at least for now. There’s a clear push to polish up both the synthesis methods and downstream reactions, nudged by demand for greener, more efficient chemical routes. Academic interest in selective aromatics and functionalized benzaldehydes picks up with each new pharmaceutical lead or high-performance material. If enzyme-driven approaches ever break through, we might see broader access to Mesitaldehyde, with a lighter environmental touch. For all the tradition behind it, this chemical keeps surprising people who spend enough time in organic labs, and I don’t see that changing anytime soon.
Mesitaldehyde doesn’t get a lot of headlines outside a chemistry classroom, but I’ve seen it turn up in more research papers and industrial projects than you might expect. Seeing that name on a reagent bottle reminds me of time spent in hot labs, frustrated and excited at once, coaxing molecules to take shapes our group hoped could lead somewhere meaningful, like a new electronic material or a breakthrough drug lead.
Mesitaldehyde is an aromatic aldehyde, built around a benzene ring with three methyl groups and an aldehyde tail. Chemists value these details not for trivia night, but because alteration offers handles for reactions. That synthetic flexibility makes mesitaldehyde a building block in the broader game of making complex organic molecules.
Take pharmaceutical research. Small tweaks on an aromatic ring can make a big difference in activity or toxicity. Mesitaldehyde steps in as a starting point to craft flavors, fragrances, and even new drugs. Its structure lets researchers shuffle around atoms and bonds safely, mapping new territory in molecular design. While mesitaldehyde itself doesn't end up on pharmacy shelves, the chemistry branching off it often seeds molecules that matter in medicine.
My own interests have turned more toward materials as years have rolled on. In the world of advanced polymers and electronic materials, starting molecules decide so much: flexibility, electrical properties, even how plastic handles sunlight. Mesitaldehyde has found a niche as a stepping stone for tailoring certain resins and specialty plastics, used in settings where regular polymers break down or underperform. Synthetic chemists value its perfect blend of bulk and reactivity, allowing subsequent steps to attach other useful groups.
Developers trying to build better OLED displays or light-harvesting components sometimes start their journey with a flask of mesitaldehyde before walking through five or ten more reactions to land on the final product.
Handling aldehydes always brings a reminder of lab safety. Even the tough old chemists who think of nothing of breathing in the faint tang of organic volatiles tend to give mesitaldehyde a little respect. Exposure limits don’t get much discussion outside the lab, but mishandling aromatic aldehydes—skin, lungs, eyes—can cause irritation or worse. Getting familiar with a chemical means knowing its risks firsthand. Good labs build responsibility into their culture, not just routine. Younger chemists need experienced folks to pass along that respect for both the molecule and the person handling it.
Folks outside the chemistry field rarely hear how these somewhat obscure building blocks shape daily life. Phones, car dashboards, heart medicines—all can trace their roots back through generations of reactions involving simple molecules like mesitaldehyde. For research to deliver its promises, it needs more than just good technique. Ethical use, honest reporting, and transparency drive trust with the public. Mesitaldehyde stands as a quiet symbol of how every chemical can be part of a web—it’s on all of us to use it wisely and talk plainly about where it can lead.
If future research keeps open lines between lab practices and community interests, mesitaldehyde keeps its place as a small, safe cog in bigger machines. Setting and following proper handling rules, monitoring releases, and educating users make real differences. What seems like a minor player matters more than most folks realize. In each experiment and product, there's an echo of hundreds of such ingredients, all deserving diligence and clear-eyed stewardship.
Mesitaldehyde sits in a small club of aromatic aldehydes known for stubborn stability and unique bulk. With its trimethyl-substituted benzene ring, it stands apart from other aldehydes. Anyone who ever stepped foot in a sophomore organic lab knows the sharp tang of benzaldehyde, but mesitaldehyde brings a different profile to the bench, one that feels far less reactive and more laid back.
You won’t spill mesitaldehyde and soak the room in vapor. I remember pulling it out in grad school: light yellow oil, thicker than you’d expect, with a heavy, nearly medicinal scent compared to the simpler ring compounds. Its melting point comes in low, staying liquid well below room temperature, which means you can pour it straight out of the bottle, unlike vanillin or traditional benzaldehyde, which tend to crystallize if you leave them too long.
A key physical trait is its resistance to oxidation. Many aldehydes darken in air, turning to acids in no time, but the crowd of methyl groups crowding the ring in mesitaldehyde keeps this process in check. That makes storage and handling easier—code for “thankfully less headache for the chem lab prep.” Pour some mesitaldehyde, and you won’t find much clinging to your gloves, thanks to its lower volatility.
Mesitaldehyde’s chemical stubbornness comes from the three methyl groups on the benzene ring. Electron-rich rings usually rev up chemical reactions, but here those methyls take up so much space that most reagents can’t get close enough for action. Classic reactions, like forming oximes or hydrazones, still work if you give them time or coax them with heat. Yet, standard nucleophiles face a challenge trying to attack the aldehyde's carbonyl carbon.
In real-world chemistry, this makes mesitaldehyde a specialty starting material. It rarely pops up in standard classroom experiments because other aldehydes give quicker results. For anyone working in research searching for selective reactivity or new ligands, this molecule offers a puzzle worth solving. The sheer crowding of substituents makes it useful for studying reaction mechanisms where other, less hindered aldehydes fail.
The stability and controlled reactivity aren’t just a chemical curiosity. In environmental labs, for instance, where trace contamination can throw off entire studies, having an aromatic aldehyde that won’t degrade in a week gives you reproducibility. In the pharmaceutical industry, aromatic aldehydes serve as building blocks for all sorts of active molecules. The robust nature of mesitaldehyde lets chemists introduce the aldehyde group exactly when they want—no surprise side products messing with the workflow.
Of course, such inertness limits its appeal in large-scale industrial settings. Speed and cost often take priority, so mesitaldehyde usually stays in niche areas. The university research bench or small-batch specialty synthesis remains its home.
For chemists hoping to unlock new potential from mesitaldehyde, the challenge lies in outsmarting the methyl crowding without resorting to extreme conditions. Catalysis and clever reagent design matter here. A few research teams in the last decade started using organometallic catalysts to coax it into new reactions, opening doors to molecules with no better route. There’s also room for green chemistry: using sustainable solvents and catalysts to make working with mesitaldehyde less taxing on both researchers and the environment.
Mesitaldehyde demonstrates how a molecule’s quirks shape the way we investigate, innovate, and sometimes battle through hardship in the lab. In the quest for new chemistry, molecules like this keep the work grounded and remind us that progress grows out of both challenge and curiosity.
Mesitaldehyde doesn’t attract as much attention as some other chemicals. You won’t see frightening headlines about it in mainstream media. Still, it belongs to the family of aromatic aldehydes, and experience has taught many of us to approach such substances with respect in the lab and in industrial use.
Anyone who’s spent time in a university lab can remember the sharp, sometimes harsh odors of these sorts of chemicals. The smell alone sometimes gives you a hint: this isn’t something to splash around. Mesitaldehyde’s structure, featuring both methyl groups and an aldehyde, sets off warning bells for folks who know how easily small molecules can find their way into the body—through skin, through lungs, through carelessness.
The available research paints a clear enough picture. Mesitaldehyde is an irritant. Aldehydes generally react with proteins and other biological molecules, which often translates to tissue irritation or more serious health effects with enough exposure. No one wants to breathe vapors or get a liquid like this on bare skin. Short-term exposure could mean watery eyes, sore throat, or a nasty rash. Long-term risks? That’s tougher to pin down. For some aromatic aldehydes, animal research has shown possible organ toxicity and even cancer risk, especially when exposure happens often or over many years.
Mesitaldehyde hasn’t been wrapped up in the same level of regulatory scrutiny as formaldehyde, which is a well-known carcinogen. That doesn’t mean it’s been proven safe—it just means research lags behind, and regulatory bodies haven’t sounded the alarms. In settings where it pops up, such as certain chemical syntheses or niche labs, safe handling practices become crucial. Protective gloves, well-ventilated workspaces, and proper storage cut down the chances of accidents.
I’ve seen what happens when shortcuts take the place of safe work habits. It takes just a single mistake—a poorly secured bottle, a missed glove—to turn a routine task into a health scare. People may get lulled by the absence of headline-making disasters, but that mindset causes more trouble in the long run. Even small, repeated exposures build up risk that experts like industrial hygienists caution against constantly.
Information gaps often exist because chemicals like mesitaldehyde don’t see wide public use, but the basics ring true: the absence of acute accidents doesn’t negate real potential for harm. Practical chemistry, at any scale, demands respect for these risks. Real-world handling experience shows that engineering controls—like fume hoods and sealed containers—are worth every penny, not just for legal compliance but to avoid the personal misery of chemical burns or lifelong allergies.
Truth is, better answers start with awareness and education. Companies and labs improve safety records not only by relying on published safety data, but by sharing stories of close calls and near misses. A well-run shop invests in real training, makes time for drills, pushes people to learn the “why” behind every warning label on a bottle. People protect one another best when everyone treats every new chemical as a potential source of harm until shown otherwise.
If mesitaldehyde crosses your path, give it the caution it deserves. Don’t gamble on what researchers might find tomorrow. Emphasizing safer substitutions or improved ventilation keeps more people comfortable, healthy, and coming back for another day of problem-solving. Safety doesn’t slow down science; it clears the path for smarter work and genuine progress.
Anyone who’s worked with chemicals in a lab or industrial setting knows safety doesn’t get paused for quick jobs or shortcuts. Mesitaldehyde stands out as a chemical that belongs on the “treat with respect” list. This aromatic aldehyde—used in organic synthesis—can bring risks if left unchecked. Over the years, I’ve seen plenty of cases where good habits made all the difference between routine work and a dangerous situation. No matter how familiar a material seems, complacency courts trouble, especially with compounds capable of irritating eyes and skin or posing inhalation hazards.
Messy shelves and mystery bottles? Not worth the risk. Mesitaldehyde lasts longer and stays safer in its original, tightly closed container. Air and light accelerate breakdown and contamination. I’ve found that stash in a cool, dry spot, well away from direct sunlight, keeps bottles in good shape for extended periods.
Fluctuating temperatures can spoil even sealed batches. Many lab veterans swear by storage below room temperature, though refrigeration isn’t always essential. The main goal isn’t some textbook-perfect environment but staying consistent and avoiding places that heat up or get damp. Any strong oxidizer nearby can spark dangerous reactions. So, keeping incompatible chemicals well apart—and labeling shelves—isn’t overkill. It’s common sense earned through experience or hard lessons.
Handling this stuff isn't about piling on layers for show. Nitrile gloves, splash-proof goggles, and a good lab coat go a long way in protecting skin and eyes. Fume hoods aren’t an optional extra—if you’re pouring or transferring, make sure the ventilation actually works. That’s not being cautious for its own sake; accidental inhalation irritates respiratory systems and sometimes brings more severe symptoms. Good ventilation means you’re not gambling with your lungs.
Spills do happen. Quick, calm cleanup matters more than heroic panic. I keep absorbent pads and waste containers nearby, so nothing lingers, and any contaminated material gets bagged and tagged for proper disposal. No draining liquid.
Reviewing safety data every now and then may feel like overkill, but regulatory requirements and recommended practices shift over time. I check labels, and if uncertainty creeps in, asking a supervisor or looking up recent guidance beats bluffing. It pays to keep up with updates from recognized organizations and experts, not just old notes in a binder.
No single rule or device alone guarantees safety. What sets safe labs and factories apart are people holding each other accountable, paying attention, and sticking to routines. Regular safety walkthroughs uncover issues before accidents brew. Small changes, like better lighting in storage areas or reminding coworkers to close containers properly, cut risk more than expensive new equipment sometimes can. Sharing real stories about close calls reminds everyone that the dangers aren’t theoretical.
Safe storage and handling demand more than memorizing guidelines. They depend on everyday decisions, honest communication, and a steady willingness to speak up if something doesn't look right. In the workplaces I trust most, everyone feels responsible—not just for their own safety, but for the whole team. That kind of culture keeps dangerous chemicals from becoming tomorrow’s headlines.
Walk into a modern laboratory or a manufacturing plant, and you’ll likely encounter chemicals most people have never heard of. Mesitaldehyde is one of those unsung molecules that supports industries, even though it rarely gets the spotlight. I’ve watched how it fits into workflows and business models, giving chemists and manufacturers more options for creativity, precision, and reliability.
Mesitaldehyde’s strongest claim to fame comes from chemical synthesis. For researchers looking to build complex organic molecules, its unique structure—three methyl groups attached to a benzaldehyde backbone—offers real advantages. In the lab, it acts as both a building block and a protective group, letting chemists piece together molecules in ways that would be far trickier without it. This kind of flexibility speeds up the process for developing new pharmaceuticals and materials, removing roadblocks that would have slowed down R&D even a decade ago.
Pharmaceuticals rely on precise reactions, and mistakes can cost months—or even years. Mesitaldehyde gives chemists a tool to create intermediates, especially those needed to build more complex drugs. It enables targeted modifications to aromatic rings, which show up so often in medicines that the market would feel its absence. For people working at the intersection of research and production, this means they see fewer failed batches, improved yield, and a smoother journey from concept to clinic.
Outside medicine, industries like plastics and polymers use mesitaldehyde in specialty applications. The electronics sector, for example, values stability and precise material properties. Mesitaldehyde makes it easier to design resins and specialty plastics that handle heat or electrical current without breaking down. Its presence, even in small quantities, can set apart a high-performance material from cheaper and less stable alternatives.
Consumers rarely think about what gives a perfume its distinctive edge or a household cleaner its pleasant scent. Mesitaldehyde sometimes serves as a precursor for aroma compounds. Its structure allows manufacturers to synthesize specific fragrance notes that would be tough to extract from natural sources. This kind of innovation keeps perfumes, soaps, and even air fresheners smelling fresh and consistent. In flavors, regulatory rules often limit direct use, so the impact is mostly indirect—manufacturers use mesitaldehyde routes to create flavoring agents for complex products.
Every industry faces pressure to improve processes and reduce waste. Mesitaldehyde is often made through routes that generate less hazardous byproduct, especially compared to older chemicals with similar functions. For companies focused on greener manufacturing, mesitaldehyde offers a way forward. Industry groups and researchers keep working to streamline these processes further. I’ve seen real progress as new catalytic systems reduce both cost and environmental impact.
Mesitaldehyde remains a niche chemical, but its influence extends beyond the factory floor. The next time you use electronics, enjoy a modern fragrance, or read about new drugs in development, you’re seeing the result of years of incremental improvements—many driven by low-profile chemicals just like this one. The continued drive for better performance, reduced waste, and safer manufacturing means mesitaldehyde will likely remain quietly essential in industries that shape our daily lives.