Some chemicals reach the lab bench and then quietly fade into obscurity, but chlorodiphenylphosphine earned its place in modern chemistry through a steady climb. Introduced to synthetic chemistry circles not long after organophosphorus compounds gained serious attention in the early twentieth century, chlorodiphenylphosphine caught the eye of researchers looking to shape molecules with phosphorus backbones. The 1940s and 1950s set the stage for wider adoption, with its unique structure—where phosphorus links to two phenyl groups and one chlorine—offering a direct route to a variety of organophosphorus derivatives. That boded well for scientists in both universities and industry, especially during a time when chemists worked with glassware and intuition rather than digital simulations and automation.
Chlorodiphenylphosphine delivers more than its name implies. Its core structure—phosphorus, two aromatic rings, and chlorine—mixes reactivity with stability. That blend makes it something of a building block for more complex molecules. It owes its continued popularity to versatility, not just because it reacts well, but also because it helps manufacturers and researchers reach targets that simpler chemicals cannot touch. The molecule can support everything from pharmaceutical syntheses to specialty polymers. Chemists favor it because it can behave as both an electrophile and a nucleophile depending on the context, offering a toolkit when adapting a route or trying to solve a persistent bottleneck.
Chlorodiphenylphosphine shows up as a colorless to pale yellow liquid, carrying a fairly distinct odor that most seasoned chemists come to recognize. It boasts a boiling point high enough to keep it manageable during typical reactions, but it won’t survive open air for long. Exposure to moisture spells trouble, as hydrolysis can run rampant and fill workspaces with harmful byproducts. The density and viscosity feel close to common organic solvents, but the real challenge comes from reactivity. Chlorodiphenylphosphine reacts with water, alcohols, and amines with sometimes startling vigor, tossing out hydrogen chloride or producing new phosphorus species in seconds. That means chemists keep it under inert gas as a standard precaution.
Quality makes a difference in any lab, and the world of chlorodiphenylphosphine is no exception. Producers test every batch for high purity, minimal moisture content, and absence of byproducts that could interfere with downstream chemistry. Labels spell out handling conditions, with most suppliers enclosing it in amber bottles or metal cans. Typical warning labels highlight the dangers of skin or eye contact, as well as the risks posed by inhaling vapors or fumes created during reaction set-ups. These technical controls offer more than legal compliance—they protect against the genuine hazards that anyone working with phosphorus-chlorine bonds must face.
Producing chlorodiphenylphosphine rarely deviates from the tried-and-true route involving phosphorus trichloride and benzene or related substrates, often in the presence of aluminum chloride as a Lewis acid catalyst. The method, simple in theory, asks for a careful hand and clear focus in practice. Reactants need to stay dry, and reactions unfold behind glass under constant watch. Temperature profiles must be monitored closely, with product distilled away from leftover reactants and purified further before anyone trusts it in a critical application. While newer methods pop up from time to time, the classic approach involving direct chlorination and controlled substitution remains a mainstay because it delivers both yield and consistency.
This compound doesn’t sit idle on the lab shelf. Chemists run dozens of distinct reactions with chlorodiphenylphosphine as a core reagent, including the production of phosphine oxides, phosphinites, and ligands for metal-catalyzed processes. Reactions with alcohols yield diphenylphosphinite esters, which later serve as intermediates in pharmaceutical or agrochemical work. Grignard reagents convert it into triphenylphosphine—another heavyweight in the organophosphorus family. The capacity to shuffle functional groups and attach phosphorus to new frameworks gives this chemical a long shelf-life in any research or manufacturing environment looking for adaptable organic synthesis tools.
Names in chemistry seldom make things simple. Chlorodiphenylphosphine goes by more than one label, including diphenylphosphinyl chloride and PDCP. Anyone reading through scientific journals or regulatory documents can find alternate spellings or abbreviations, but the core substance remains the same. Recognizing synonyms matters for safety data, chemical sourcing, and interpretation of research literature. No matter the brand or country of supply, those in the industry keep a mental shortlist of accepted product names to ensure nothing gets mixed up on the bench or in inventory systems.
Handling chlorodiphenylphosphine does not equate to dealing with everyday solvents or mild laboratory acids. Splash risks send many to emergency showers or fume hoods, and the corrosive, toxic nature leaves little room for error. Training focuses on airtight storage, nitrogen or argon blankets for bulk quantities, and chemical spill kits stationed within arm’s reach. Personal protective equipment moves from afterthought to essential—neoprene gloves, goggles, lab coats, and face shields all feature in experienced hands. Facilities working with chlorodiphenylphosphine design engineering controls because ventilation alone cannot safeguard against accidental exposure or aerosol formation.
Diverse industries draw on what chlorodiphenylphosphine offers. Agrochemical firms use it for pesticide and herbicide intermediates, while pharmaceutical researchers deploy it for core transformations unreachable with carbon-based analogues. Catalysis experts transform it into ligands used in metal-based cross-coupling chemistry. Advanced material scientists employ organophosphorus derivatives in fire retardants and specialty polymers. Even electronics benefit from niche applications in semiconductor processing where phosphorus compounds act as dopants or etchants. Anyone walking through a functional chemical plant or development lab will likely spot this molecule or its products sooner or later.
Every few years, new research unveils wider uses or less toxic derivatives of chlorodiphenylphosphine. Academic groups and industrial R&D teams investigate synthetic routes to reduce environmental impact or side-product formation. Some chase after greener oxidants or milder chlorination techniques, while others engineer catalysts built on the diphenylphosphine framework. Analytical scientists value it as a source of phosphorus-31 NMR references. The pipeline from molecule design to market product runs through extensive in-house analysis and process verification, demanding both innovation and rigor.
Chlorodiphenylphosphine never shies away from controversy when it comes to toxicity. Researchers document its acute toxicity in animal tests, with particular attention paid to inhalation and dermal absorption risks. Once inhaled or absorbed, the compound can cause respiratory distress, severe skin burns, or eye injury. Data from animal studies drive regulatory controls for workplace exposure, but much remains to be done regarding long-term environmental fate and breakdown products. Chemists treat any spill or accidental release with utmost seriousness. History has shown that even seasoned professionals must respect the immediate and long-term risks, using air monitors, detector tubes, and rigorous waste protocols at all stages of use.
Chlorodiphenylphosphine likely has more in store for the scientific community. As green chemistry gains steam, pressure mounts to slash hazardous waste and improve overall safety. Efforts to develop less toxic analogs, recycle phosphorus waste, or capture and neutralize hazardous byproducts before discharge are already underway in many labs. Advances in automation and process safety look set to reduce human exposure further. Intellectual curiosity, paired with tighter regulation, will keep pushing for safer, more efficient uses of this molecule in new domains. Watching the next decade unfold will reveal not just where chlorodiphenylphosphine fits, but how it inspires chemists to work smarter and safer across many fields.
Few people outside chemistry labs ever hear about chlorodiphenylphosphine, but this sharp-smelling combination of phosphorus and chlorine shapes an entire corner of modern industry. I used to think chemicals with tricky names stayed locked in a lab, sealed behind big glass doors and blue-gloved hands. Chlorodiphenylphosphine has a long reach, though. Its traces thread through pharmaceuticals, plastic manufacturing, and agrochemicals, quietly setting the stage for some of the tools and conveniences people take for granted.
Walk into a chemistry lab and the story is clear: progress starts with basic building blocks. Chlorodiphenylphosphine acts as one of those builders, especially for making new organophosphorus compounds. These don’t end up on store shelves themselves, but boil them down, and you find their fingerprints on products like flame retardants, special plastics, and even some medicines.
You might be surprised at how many products depend on chemicals like this—not only industrial supplies but also consumer goods. A single reaction somewhere in a factory can ripple all the way to the safety coating on a wire or a specialized resin in construction. Sometimes, I’ve seen the value of this approach firsthand. Grad school meant endless hours fiddling with reaction pathways, looking for something stable, reusable, and safe. Chlorodiphenylphosphine stuck around because it does that work efficiently, allowing chemists to add phosphorus in the right place, often with fewer unwanted side products.
The demand for specialized chemicals keeps climbing. Smart agriculture needs new crop protection tools that work without causing harm to people or nature. Chlorodiphenylphosphine feeds into the creation of these tools—seed treatments, weed management, and specialty fertilizers often come from the compounds it helps build.
Pharmaceuticals, too, lean on its power. The path from a raw material to a new pill on the pharmacy shelf can stretch for years and bend through a maze of regulations. Starting with materials like chlorodiphenylphosphine saves time and resources. In the lab, reactions with this chemical can be easier to control, meaning fewer failed batches and more predictable results. That keeps research costs down and helps speed promising drugs to patients waiting on hope.
Handling chlorodiphenylphosphine doesn’t feel like the chemistry sets from childhood science class. This chemical burns if it touches skin, and its vapors sting the nose and lungs. Accidents can get serious in a hurry. Decades of experience show the importance of solid safety systems—ventilation, storage rules, and proper training. There's no way around the risk; the only way forward is honest, steady attention to how people interact with the material. People working with it deserve equipment that keeps air clean and hands safe, as well as clear instructions from employers who take chemical safety seriously.
Some companies have already started questioning traditional chemical routes. Newer, greener reactions might eventually edge out older, harsher players like chlorodiphenylphosphine. Today, though, replacing its role would mean reshuffling a lot of carefully tuned processes, especially in pharma and advanced materials. The path to safer, less toxic chemistry won’t happen overnight, but sharing experiences and supporting research into alternatives opens real possibilities.
People may never hear about chlorodiphenylphosphine at the kitchen table. That quietness doesn't lessen its impact. Behind the scenes, the choices chemists make—including how and why they use chemicals like this—echo farther than most folks could imagine.
Anyone who’s spent time working with chemical reagents picks up a deep respect for personal safety. I’ve seen what carelessness can do, not just to a person, but to the whole environment around them. Chlorodiphenylphosphine, with its sharp reactivity and health hazards, counts among those chemicals that demand extra vigilance. This compound brings a heated urgency to safety conversations, and for good reason: Even a small mistake can lead to burns, inhalation injuries, or toxic spills.
One can never underestimate the value of solid personal protective equipment. I still remember my first time using a fume hood for reagents like chlorodiphenylphosphine. Gloves and lab coats made me feel prepared, but it’s the goggles and fully sealed face shields that catch the real threat here. Spattering is not just a theoretical risk; it happens. The compound reacts with water vigorously, meaning sweat, humidity, or stray droplets put hands and arms in direct danger.
Without a fitted respirator, fumes may travel up into your lungs before you realize the leak. Chlorodiphenylphosphine releases toxic gases, sometimes with a delayed reaction, so you want that mask on before you even open the bottle.
People forget just how dangerous poor storage can be. I’ve seen bottles left uncapped for a moment catch enough humidity to start a chemical scare. Only keep this compound in tightly sealed, chemically-resistant containers. Keep it far from water sources — even cleaning up a spill becomes a dangerous process if the area isn’t bone-dry. Refrigerators and cabinets built for storing corrosives are the minimum standard, never a luxury.
No one plans to drop a flask, but it happens. Spill kits need to be within arm’s reach and stocked with the right neutralizers. Sometimes, labs cut corners and rely on generic kits; specialty chemicals like chlorodiphenylphosphine need their own neutralization protocols, often using non-water-based absorbents. Training is not just about reviewing slides. Getting everyone in the lab to practice with simulated spills builds a culture where people know what to reach for… before panic decides for them.
A fume hood should always be the frontline defense. A workspace without effective ventilation turns into a trap. My experience tells me that people often ignore alarms or sensor warnings, treating them like background noise, but gases from chlorodiphenylphosphine move fast. Investing in air quality monitors means no one has to guess about exposure or cross their fingers after every experiment.
There’s no shortcut to hands-on education and a safety-first mindset. Trust in science depends on each lab worker returning home healthy. Real safety commitment means ongoing training, upgraded protective gear, and refusal to normalize routine shortcuts. Reviewing near-misses and sharing stories about accidents make dangers feel real, not just theoretical. Facing chlorodiphenylphosphine, respect becomes a daily practice, and that vigilance is what keeps labs running and people out of harm’s way.
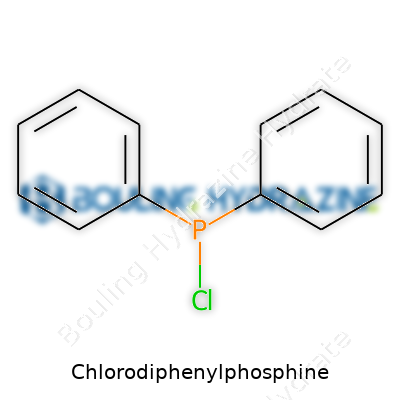
Chlorodiphenylphosphine carries the chemical formula C12H10PCl. Looking at that mix of carbon, hydrogen, phosphorus, and chlorine, some see another routine molecule. Those who deal with this compound day in and day out know it’s not some faceless ingredient. This stuff matters—a lot—especially for chemists working on organophosphorus chemistry. The backbone of the molecule lines up with two phenyl rings (benzene rings, for those who haven’t dusted off a high school textbook in a while) bonded to the central phosphorus atom. Then phosphorus links up with a chlorine atom: simple at a glance, but powerful in purpose.
I learned early on, working in a university lab, that text-book chemistry formulas can’t always capture the real implications. The structure here—two bulky aromatic rings attached to phosphorus—gives the molecule a distinct attitude. These rings create both a physical barrier and a unique electronic environment around the phosphorus center, which makes chlorodiphenylphosphine especially useful for building more complex molecules. The presence of that chlorine isn’t just decorative. Chlorine turns this compound into a solid starting point for making pesticides, flame retardants, and specialty chemicals.
Demand for smart chemistry grew over the past decades. As regulations on many chemicals tightened, safer and more efficient ways to create new phosphorus compounds rose in priority. Chlorodiphenylphosphine became a workhorse for making certain herbicides and pharmaceuticals. For example, the chlorine atom can get swapped out in reactions, which enables scientists to attach all sorts of new groups onto the phosphorus. This versatility drew in researchers trying to improve everything from crop protection to materials used in electronics.
Science journals and regulatory agencies both track the roles of compounds like this. According to the EPA, careful handling and storage are necessary because of its reactivity, especially upon contact with water—it produces hydrochloric acid and diphenylphosphine oxide in those conditions. Mishandling can mean exposure to corrosive vapors, and that’s not just a safety slogan. Once, a careless moment with glassware showed me how quickly an ordinary day can turn unpleasant.
Most discussions about laboratory chemicals skip over their environmental fate. Chlorodiphenylphosphine doesn’t get much press, yet its breakdown products can create concerns. Better storage and handling rules help, yet there’s room for greener chemistry. Catalysts that perform similar synthetic tricks, without the legacy of hazardous waste, are out there. Investment in research that emphasizes sustainability, encourages recycling of by-products, and reduces the need for harsh reagents strengthens the case for responsible progress.
Chemistry headquarters for green manufacturing shouldn’t only focus on end-use. Real progress happens when the supply chain, from raw materials to disposal or recycling, falls under scrutiny. Students in chemistry deserve to learn both the detailed structures and the broad context. Every time researchers develop new methods to substitute hazardous components, they reinforce responsible science.
Identifying chlorodiphenylphosphine by just its formula doesn’t give the full picture. Chemical structure sketches usually show the phosphorus atom at the center, linked by a single bond to a chlorine atom on one side and to two benzene rings on the other sides. This specific arrangement unlocks its wide range of chemical possibilities. It also places responsibility squarely in the hands of those working with it, making chemistry innovation something more than a paper exercise.
Anyone who works in labs or chemical storage knows some substances feel like they're just waiting for the chance to ruin your week. Chlorodiphenylphosphine lands squarely on that list. The label on the drum might list it as a “reactive organophosphorus compound,” but the real lesson only arrives for folks who’ve handled it up close. From what I've seen, this isn’t the kind of bottle you want forgotten in a cupboard or left sitting open at the edge of a fume hood.
This chemical reacts quickly with moisture in the air, and that reaction can release dangerous fumes. Not everyone has seen what happens when a container leaks or sweats in a humid storeroom, but those who have don’t forget it. That vapor is more than just a sour smell. Inhaling it can sting eyes and lungs, and skin contact carries its own set of health worries. A moment’s carelessness can mean scrambling for the eye wash or worse, so talking about safe storage isn’t an exercise in bureaucracy.
Glass bottles with tight seals work best, tucked inside sturdy metal cans for an extra layer of protection. That double-layer, glass inside metal, doesn’t just sound fancy — it limits both leaks and accidental bumps. For folks who skip this step, a single dent, or drop can spell a big mess. Store these bottles in a dry place, far from sinks, wash stations, or any source of water. With the way this compound reacts, even a little splash is too much.
Temperature swings bring their own troubles. Keeping things cool and steady — not freezing, not toasty, just away from heat and sunlight — helps avoid surprise pressure build-up. I've seen labels peel off or go unreadable in steamy rooms, which is an easy way to forget what’s inside. Labeled containers, always, with clear writing anyone on shift can read. That prevents confusion when time matters.
Warehouses sometimes store this compound by the drum. It’s smart to keep incompatible chemicals away, especially acids and bases, since a spill could double the danger. Out-of-the-way chemical storage rooms with spill trays and proper ventilation keep minor slip-ups from becoming emergencies. I’ve watched as simple precautions — absorbent mats, secondary containment tanks — kept small leaks from spreading. That saved a lot of grief when it counted.
Relying on a once-a-year inspection won't cut it. A smarter approach is regular walkthroughs by experienced eyes, especially just before weekends or holidays. Written storage procedures, real training for new workers, and a culture where people double-check each other make sure this stuff isn’t forgotten or misplaced.
Forget the glossy how-to posters — real stories and peer-to-peer knowledge stick with people better. Incentives for strong housekeeping, routine label checks, and making sure every team member can spot a risky situation beat any policy binder I’ve ever read. Storage habits change through practical experience and honest talk, not through scare tactics.
Chlorodiphenylphosphine deserves respect, not fear. Proper storage isn’t a hassle: it’s the daily work that keeps everyone healthy, and it saves time down the road. Every safe shift is proof that straightforward steps work best.
Chlorodiphenylphosphine doesn’t grab headlines, but in the chemical world, it commands respect. This clear, colorless to yellow liquid often turns up in custom synthesis labs, specialty chemical facilities, and sometimes in academic research. Trouble is, with that pungent smell comes more than just inconvenience: it means danger on several fronts. Breathing in fumes can hit you fast—irritation, coughing, and sometimes chest pain show up almost right away. Even a short encounter stings eyes and burns nasal passages. Skin contact leads to itching, redness, and burns in heavy exposures. Any splash reaching your eyes risks permanent damage.
Corrosive is not just a label—it means real tissue damage. This chemical produces toxic gases once it runs into water or moisture, forming hydrochloric acid and some nasty organophosphorus fumes. That’s a serious hazard for anyone carrying out even a routine transfer, let alone during a spill. Reactive incidents have left people scarred for life, and some unlucky workers in confined spaces paid with their health after breathing vapors from a leaky container.
You never forget being in a space that suddenly reeks of burning garlic—a sign of organophosphorus exposure—while alarms shriek and everyone’s scrambling for eyewash and fresh air. Acting fast is not some hypothetical. An exposed colleague who reaches an eyewash in under twenty seconds will likely keep their vision. Delaying even a minute can mean partial, sometimes total, loss of sight.
For me, seeing this play out in real time erased any doubt about the value of repeated drills and fresh emergency gear. Teams working with chemicals like chlorodiphenylphosphine need systems that actually work. This begins with clear labeling and airtight storage, but real preparedness includes proper personal protective equipment and being able to reach an emergency shower with your eyes closed—literally.
If vapors have gotten into someone’s lungs, get them into fresh air. Forget debating; just move. Remove contaminated gear right away, as clothing holds onto vapors and liquid, making things worse. Skin contact means flushing the area with water for at least 15 minutes. That’s not a guess or a nice idea—rushing the washout often leads to burns developing later. If the chemical splashes into the eyes, the only right move is heading to an eyewash and letting a stream of clean water roll over the eyes non-stop. Don’t waste time trying to “wash out” with a bottle, and do not delay seeking professional medical help afterward.
Swallowing this chemical rarely happens, but if someone does, rushing for water and getting medical attention is vital; do not cause vomiting. Rescuers run a risk too, so self-protection remains non-negotiable. Protective gloves, splash-resistant goggles, and lab coats go from precaution to necessity.
Lab safety culture remains the best antidote to chemical complacency. Having eyewash stations and showers within reach changes outcomes. Training sessions run by people who’ve seen real-world accidents—a seasoned safety expert, a lab manager who still has scars—drives home what textbook advice never does. Investing in proper storage and penning up clear emergency procedures isn’t about obeying a rulebook. It’s about bringing everyone home at the end of the day.
Working with chlorodiphenylphosphine will always come with risk. Resisting shortcuts and refusing to accept “it hasn’t happened yet” as a safety plan, that’s how labs and workers avoid showing up in accident reports or news stories. The right habits and gear tip the odds in your favor every time.