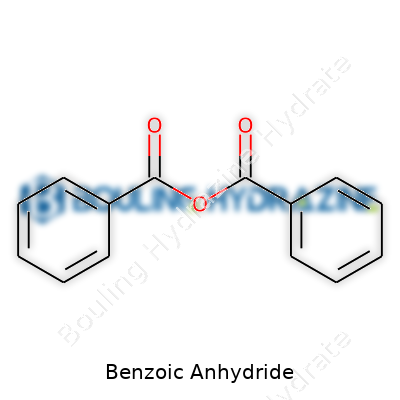
Benzoic anhydride hardly tops the list of household chemical names, but even the least understood compounds can play important roles in chemical history. Early curiosity about benzoic acids in the nineteenth century provided a pathway, as chemists started exploring how organic acids combine and dehydrate. As laboratories grew increasingly sophisticated, benzoic anhydride stood out for its ability to bring two benzoic acid molecules together by shedding a molecule of water. The process didn’t require rare ingredients, just skill and careful temperature control. Decades ago, books described its white, powdery look and pungent odor, noting that its crystalline nature made it stable for scientific storage even with basic glassware. Over generations, its laboratory value persisted among those who appreciate clear, well-behaved reagents with dependable reactions.
Anyone who has encountered benzoic anhydride remembers how it looks and behaves. The crystal powder tends to catch light, showing off a dry white color. Tossing a pinch on the open palm, it may clump slightly due to its fine grain, and a sharp scent lingers. At standard temperature, it holds form, melting around 42 degrees Celsius—barely above body temperature. Its molecular weight raises eyebrows among undergraduates, just hefty enough to command respect yet modest compared with bulky organics. It reacts quickly in the presence of water, eager to hydrolyze back to benzoic acid, burning off the characteristic anhydride odor. As a molecular building block, it shows its readiness to acylate and react without complex catalysts, making it unique compared with other traditional acid derivatives in a well-equipped lab.
On store shelves or laboratory inventories, benzoic anhydride demands a clear, accurate label, both for safe handling and trade. Cas numbers and molecular formulas produce eye rolls for some, but for someone mixing potentially volatile reactants, nothing beats simple clarity—knowing you have exactly what the label indicates. Transparent technical specifications guide safe use: purity should cross the 98% mark for bench chemistry to avoid unpredictable results, and the product shouldn’t contain traces of heavy metals, which could sabotage sensitive syntheses. Regulatory compliance isn’t just paperwork. Some jurisdictions require hazard warning symbols, especially as inhalation or contact can irritate. Consistent, honest disclosure preserves trust between supplier, scientist, and regulator.
Traditional preparation hinges on dehydration, using strong acylating agents and precise temperature ranges. The process usually begins with high-purity benzoic acid, with phosphorus pentachloride or thionyl chloride stripping away water, pushing the reaction forward. I’ve watched chemists curse over slow crystallization or incomplete conversion in poorly ventilated fume hoods, as even tiny environmental changes swing the outcome. Yield varies depending on skill and patience; too much heat and the product darkens, too little and excess reactant lingers. Scaling up the method for industrial runs ramps up the risk and cost due to the need for careful temperature controls and containment of acidic vapors. Every synthetic chemist feels a tinge of pride pulling crisp, dry benzoic anhydride from a freshly charged flask.
On the benchtop, benzoic anhydride’s acylating power unlocks dozens of synthetic options. If a researcher wants esters or amides with a phenyl background, this compound steps in—no need for exotic conditions. For example, combining it with an amine streamlines the synthesis of N-benzoyl derivatives, whether in pharmaceutical or polymer development. Its willingness to react with nucleophiles makes it a versatile friend for those who dislike fiddling with less reactive, moisture-sensitive reagents. Sophisticated users can swap out phenyl groups for more elaborate work, testing limits and diversifying the spectrum of benzoic analogs. In the hands of a creative chemist, modification isn’t just possible—it’s expected.
Many names chase benzoic anhydride around the world: dibenzoyl oxide, benzenecarboxylic anhydride, and assorted trade synonyms. Each version enters the literature for technical precision or regulatory tone. Yet, at heart, it’s the same chemical structure at work in decades-old organic textbooks and contemporary online catalogs. Those who recognize its synonyms find a subtle pride in this specialized vocabulary—a way of distinguishing professional expertise from general conversation.
Every compound has a reputation for safety—or risk—earned through repeated use and shared stories. Benzoic anhydride deserves real caution. It irritates skin and eyes, and its vapors bite at the throat. My first exposure without gloves reminded me with a sharp tingling discomfort how careless handling can ruin a whole afternoon’s work. Institutions mandate fume hoods for good reason: small procedural shortcuts can expose users to corrosive dust or vapor. Standard protocols recommend splash-proof goggles, gloves, and long sleeves. Even careful transfer between jars can result in small puffs of dust, so training and respect for the compound prove essential. Following guidelines isn’t busywork; every story of a burned hand or red eyes underlines that lesson.
Though overshadowed by much larger industrial chemicals, benzoic anhydride’s strengths shine in specialized applications. Academic research teams use it to protect functional groups, facilitate organic synthesis, or study acylation mechanisms in educational settings. It’s no stranger to advanced pharmaceutical research, where modifying molecules with benzoyl groups tailors the properties of active compounds. Occasionally, it slides into polymer chemistry, where controlling molecular architecture requires both reactive and stable intermediates. Most of all, its role isn’t about volume but about precision—offering a straightforward route to test molecular hypotheses without adding unnecessary complexity to reaction schemes.
Over the past twenty years, shifts in organic synthesis created opportunities for benzoic anhydride to find new roles. Green chemistry urges reevaluation of traditional pathways, and new methods develop that reduce waste by milder dehydrating agents or even catalytic alternatives that cut energy usage and dangerous byproducts. I’ve seen research papers focus less on the compound itself and more on what it enables—its position as a flexible intermediate launches entire explorations of novel pharmaceuticals, fine chemicals, and advanced polymers. The push for sustainable, scalable chemistry brings renewed energy to revisiting how it’s made and handled, including recyclable catalysts and solvent-minimized protocols. Each innovation in this sphere hinges on balancing tradition with pressing environmental and efficiency demands.
Benzoic anhydride demands sober respect from those who use it regularly. Inhalation or accidental exposure leads to irritation and discomfort, so research into alternative preparation and safer handling grows in importance. Regulatory agencies periodically review the compound’s toxicity data, updating occupational exposure standards to match new findings. New studies on metabolic fate and potential carcinogenicity keep the industry alert but so far, risk remains manageable when guidelines are honored. For those in the research trenches, toxicity isn't abstract—it’s a daily calculation of benefit versus risk, shaping training, equipment choices, and emergency planning. Practices like closed transfer systems, improved ventilation, and personal monitoring continue to push safe use forward.
Looking ahead, benzoic anhydride’s future depends on both scientific creativity and regulatory clarity. As the world demands greener synthesis with reduced reliance on harsh reagents and risky intermediates, this compound’s reactivity offers promise balanced against safety and sustainability imperatives. Ongoing exploration into catalytic alternatives, scalable continuous processes, and novel uses in medicinal chemistry may catapult it into broader relevance. Many compounds fade into the background as fashions and markets shift, but benzoic anhydride’s proven track record and reliable versatility keep it relevant for anyone who values dependable results on the road to fresh discoveries. The challenge now lands with today’s generation of chemists to reimagine established compounds for tomorrow’s needs, building on the lessons and stories handed down over years of shared laboratory tradition.
Ask most folks on the street about benzoic anhydride, and you’ll probably get some blank stares. Still, this compound sits quietly behind the scenes, driving a surprising number of industrial and laboratory processes. It’s a white, crystalline solid—not much to look at, but it packs some punch when chemists want to get things done. Its main strength comes from driving reactions that swap out groups in molecules. Nature doesn’t often make benzoic anhydride, so people cook it up in factories.
Chemists often reach for benzoic anhydride because it works fast and efficiently. It acts as a dehydrating agent, which means it pulls water out of another chemical system. If you think back to high school chemistry, removing water from a reaction isn’t just busywork—it changes the entire course of the mix, letting us build bigger, more useful molecules. People use it when making things like esters or benzoic acid derivatives. If something smells fruity in a perfume or carries a specific medicinal property, there’s a fair chance the pathway included an acid anhydride at some step.
It’s tempting to dismiss these building-block chemicals as “just for the lab,” but they end up impacting our daily routines. Take certain plastics, resins, and dyes—benzoic anhydride plays a role in churning out the raw materials for these products. Producers have leaned on it because it delivers clean results and often makes fewer byproducts. In the pharmaceuticals world, chemists create intermediates with its help, paving the way for medicines to reach your pharmacy shelf.
Growing up in a family that ran a paint supply shop, I learned that even products we rarely think about—adhesives, protective coatings, specialty paints—can trace part of their family tree to heavy-duty chemicals like this one. Its influence runs deeper than we give it credit for, from the bottle cap sealant that keeps drinks fresh to the glossy finish on posters.
Not everything about benzoic anhydride exudes promise. Working with it takes training and care because it can irritate skin, eyes, and airways. I remember stories from friends in industrial settings where safety protocols were non-negotiable—not just for the sake of workers, but to keep communities safe too. Regulatory groups set limits and use tough guidelines because some sloppy handling can mean environmental issues or harm to health. Raw chemical ingredients often earn a bad reputation precisely because their risks get overlooked in the rush for efficiency.
Education stands out as one solution—companies and institutions simply can’t afford to underplay the risk. Training workers and teaching students about safe handling tips makes a real difference. It’s on manufacturers to keep an eye on new research that promises safer alternatives or tweaks. Over the years, I’ve watched industry groups encourage transparency about supply chains and chemical origins, which helps keep everyone honest. Setting up safer processes, looking for greener options, and using protective equipment move this chemical from “hazard” back to “essential tool.”
Benzoic anhydride isn't going anywhere soon. Its reputation continues to shift as the public learns more about what really goes into making modern life function. When producers get creative, blend responsibility with innovation, and put people first, even something as humble as a white powder gains purpose far outside the lab bench.
Benzoic anhydride rarely appears on the average person’s radar, but in labs and chemical storage rooms, it often sits on shelves or inside cabinets without much fanfare. The dull name masks how questions about its storage open bigger conversations about safety, responsibility, and habits that either prevent or invite trouble.
Benzoic anhydride reacts strongly with moisture and a handful of other chemicals, so storing it with a casual attitude can cause headaches—sometimes literally. Every seasoned chemist has stories about the aftermath of leaky lids or careless placement near incompatible substances. An accidental spill might sound like an inconvenience, but real incidents bring lung irritation, eye discomfort, and a scramble for the safety shower. Avoiding that starts with a straightforward rule: keep benzoic anhydride in airtight containers, far away from water sources or humid places.
I’ve worked in research spaces where policies stressed this point, yet lapses creep in whenever colleagues get complacent. Clear container labeling helps, but only if users read the labels and respect what’s at stake. Even a small amount of condensation can trigger unwanted chemical reactions, sometimes damaging other stored reagents or lab equipment.
Closed cabinets, especially those designed for corrosive or reactive chemicals, often house benzoic anhydride. Locking the cabinet limits access to those who understand the risks, and putting the material on a dedicated shelf, clearly separated from bases, strong acids, and oxidizing agents, adds a layer of protection.
Don’t neglect the environment of the storage area itself. A cool, dry spot may sound like common sense, but climate controls matter. I’ve watched summer humidity wreck entire stocks of sensitive chemicals. Regular checks—sometimes as basic as tapping the container or peeking at any signs of clumping—help catch issues before they spiral. If crystalline powders cake together or discolor, air or moisture might have found their way in.
Ignoring proper storage isn’t just an internal problem. Local fire codes, workplace safety laws, and environmental protection rules draw explicit lines around storing reactive organic anhydrides. Fines and enforcement are real possibilities for labs that cut corners or treat storage as an afterthought.
At schools and research institutions, training helps set the tone, but it’s regular, routine habits that make a difference. I remember a student, fresh from a safety course, pointing out a misplaced bottle on a crowded bench. That reminder led to a broader review and fix—showing that a culture of speaking up can do more for safety than any single policy.
Good storage practices demand more than occasional attention. Integrating checks into weekly routines, using clear labels, and providing meaningful training create a framework that shrinks the odds of mishaps. An investment in quality containers makes a difference, so don’t skimp to save a few dollars where airtight seals matter. For anyone working around chemicals, it’s not about paranoia. It’s about respecting the material and realizing that small changes in storage can save time, resources, and maybe even someone’s well-being.
Benzoic anhydride comes up in chemistry labs more than you might think. Its role in creating other chemicals makes it valuable for research, but there’s a reason it shows up on lists of hazardous substances. This chemical can irritate skin, eyes, and even the airways. Getting educated about how to work with it safely, instead of just following a checklist, helps everyone go home healthy.
Nobody walks into a lab hoping to learn the hard way about chemical burns or allergies. During my graduate years, I saw classmates getting rashes because gloves didn’t fit right or someone rushed through cleanup. Mistakes happened quickly and sometimes they lingered a bit longer than anyone expected. Simple steps, like checking the gloves and not skipping the goggles, kept half the trouble away.
Benzoic anhydride reacts with water; it doesn’t just sit there. All it takes is some spilled coffee or a wet countertop, and suddenly the air smells sharp. That’s benzoic acid forming, and if you breathe it in, your nose tells you fast. Good ventilation always makes a difference. If your workspace has fume hoods, using them should feel as automatic as putting on your seatbelt. For those in smaller setups, portable exhaust fans work as a backup; you just want to avoid breathing in the fumes.
Regular nitrile gloves stop most chemicals, but benzoic anhydride can push through after a while. Checking your gloves for even tiny rips becomes a habit. Changing out gloves during longer sessions pays off, even if it slows the day down a bit. I saw clear goggles turn cloudy from a splash, but the person’s eyes didn’t. Stories like that made personal protective gear a no-brainer for everyone after.
Some folks want to work fast, maybe with short sleeves or only half their gear. That’s where burns start. Covering up with a proper lab coat and closed shoes, even if you’re just “doing a quick transfer,” has saved me from explaining burns more than once. Simple choices here turn into long-term safety.
Keeping benzoic anhydride dry is step one. Moisture doesn’t just ruin the chemical; it can release harmful fumes. Sealing the bottle tight and storing it with other reactive chemicals off the main bench reduces headaches. Easy access matters, but so does avoiding unnecessary spills. At one point, a forgotten open bottle filled a storage closet with nose-burning vapor. Good labeling and regular checks set a higher standard than any policy document.
Disposing of leftover benzoic anhydride takes more than pouring it down the sink. Lab teams should have a waste protocol for corrosive and hazardous materials, which often involves properly labeled containers and scheduled pickups. Every team member benefits from a clear system; guessing and making up solutions on the fly only causes trouble later.
No piece of paper or online training replaces paying attention in the lab and learning from people who’ve handled these chemicals before. Simple habits—using the right gear, respecting ventilation, labeling bottles, and cleaning up carefully—go further than most realize. The risks with benzoic anhydride are real, but with a safety-first mindset, nobody has to learn the hard way.
Someone who works with lab chemicals ends up fielding a lot of basic questions. A common one is about whether benzoic anhydride dissolves in water. On the surface, the answer is pretty straightforward: no, benzoic anhydride barely dissolves in water at all. That detail might seem minor, but there’s a whole conversation about why—and what it represents for chemistry in daily life.
Benzoic anhydride stands as an example of an organic compound with hydrophobic tendencies. Structurally, it’s not friendly with water molecules, which means water molecules can’t surround and pull it apart easily. Water plays favorites; it prefers molecules that can make hydrogen bonds, like alcohols or sugars. The anhydride, on the other hand, sits there almost ignoring water completely. Stir it, shake it, heat it—a few molecules might sneak into the solution, but that’s about it.
That little fact changes how a chemist uses benzoic anhydride. In school, students sometimes think chemicals just mix together if you want them to. In practice, you need the right solvent. Acetone and ether dissolve benzoic anhydride fine. Water leaves it mostly untouched. The molecule keeps its distance, which matters if you try to run a reaction or purify a product.
Water-insoluble chemicals ask for extra caution. If a spill happens, you won’t be able to just flush it away. Instead, you need to trap it with an absorbent, sweep it up, and dispose of it properly. Over the years, I’ve seen plenty of students treat all powdered chemicals as if water will take care of them. It doesn’t always work that way, and that’s where people get into trouble with waste or exposure. Responsible disposal starts with knowing solubility. That’s basic, but you’d be amazed how often it’s overlooked.
From a practical standpoint, insolubility in water also affects industrial and laboratory processes. In some reactions, you want your reagent to stay separate, so it reacts only when you introduce the right co-reactant or solvent. Benzoic anhydride finds its way into synthesis for this very reason—it reacts with alcohols or amines once you add them in an organic solvent, often making esters or amides that show up in fragrance, pharmaceutical, and polymer work. Knowing not to use water as the medium avoids ruined batches, wasted time, and messy outcomes.
Basic chemistry lessons sometimes get skipped in the rush to learn “big ideas.” Skipping the simple facts, like solubility, undercuts real safety and efficiency. Making sure students and workers understand this helps keep everyone safe and avoids chemical mishaps. In my own teaching and lab mentoring, I drill into newcomers that the solubility chart isn’t just a piece of paper—it predicts whether something washes away, floats, or sits stubborn at the bottom of a beaker.
In a wider sense, promoting hands-on learning about physical properties can help close the gap between classroom instruction and real-world practice. I believe better training, clearer labels on chemicals, and more built-in reminders about the quirks of substances like benzoic anhydride will keep labs safer and more productive. In the end, chemistry is hands-on work. Paying attention to these details matters—one lesson at a time.
People who spend time in a chemistry lab come across all sorts of formulas that stick in the mind, but not everyone stops to consider the value behind a name like Benzoic Anhydride. Written as C14H10O3, this compound may not ring bells for most folks outside the science world, but it plays a steady role in the background where synthetic chemistry takes front stage.
Benzoic Anhydride forms when two molecules of benzoic acid lose a molecule of water and join forces. This structure, with its pair of benzene rings linked by a bridging oxygen, looks simple enough on paper. But the real interest comes from how this arrangement works in practice.
Chemists rely on substances like Benzoic Anhydride for its usefulness in acylation reactions, which lay the foundation for making various drugs, dyes, and fragrances. For anyone who's ever opened a bottle of aspirin or picked up a skincare product, the history behind molecules like this often sits in the production chain.
Naming chemicals and jotting down formulas may seem like tasks for exams or textbooks. In reality, these formulas underpin real safety practices and decisions. Knowing the chemical makeup of Benzoic Anhydride warns technicians and researchers about its reactivity, especially around water and strong bases or acids. Mixing chemicals without understanding them leads to accidents—there’s no academic exercise in that.
The fact that Benzoic Anhydride isn’t as common as regular benzoic acid means many students and workers won't see it outside more advanced labs. That’s not a bad thing; specialized chemicals should stay where they’re needed, with people trained to recognize their quirks and hazards.
Over the years, tighter chemical safety rules helped steer labs toward safer practices. Benzoic Anhydride, with its reactive nature, calls for gloves, ventilation, and a clear plan for spills. Respect for these guidelines reflects a bigger lesson; understanding chemistry means taking responsibility for the risks, not just the rewards.
Some governments focus on stricter labeling and handling, recognizing that risks come not just from the initial reaction, but also from storage and disposal. With growing attention on green chemistry, researchers look for ways to use alternatives when possible, cut down on hazardous waste, and never lose sight of the human factor behind every flask and beaker.
Solving real problems in chemistry means more than memorizing formulas. The story behind Benzoic Anhydride—how it’s made, used, and treated at the end of its life—touches on communication, training, and responsibility. Forging connections between lab processes and everyday safety, professionals gain a deeper respect for the field and for the people who keep it running smoothly.
Next time someone scribbles C14H10O3 on a whiteboard, there’s more to it than numbers and letters. It’s a reminder of the progress made in chemistry, the value of expertise, and the balance between innovation and safety.