People have chased after new syntheses and molecules as soon as chemistry crossed into the industrial era. Historical records on derivatives like 2-Dimethylamino-1,3-dithiocyanatopropane are full of small experiments and unique detours in chemical research. Years ago, sulfur and nitrogen compounds caught scientists’ attention because they held promise as building blocks for all sorts of applications. Back in the mid-20th century, researchers started pushing past the standard organic templates, looking for ways to anchor sulfur and amino groups to create tools for medicinal and industrial chemistry. Many chemists spent long hours hunting for ways to swap in or attach specific functional groups, always hunting for properties that would unlock something new in reactivity, binding, or flexibility. Research journals from decades past show a scramble for patenting new heterocyclic and chain structures, which led straight to compounds like this one. The early days were full of trial and error, but the promise of finding selectively reactive or biologically active compounds kept folks at the bench well past midnight.
2-Dimethylamino-1,3-dithiocyanatopropane packs a punch with both its structure and its chemistry. On paper, it looks like any other combination of carbon, nitrogen, and sulfur, but it hides plenty of nuanced traits. The dimethylamino group—two methyls clipped to an amino—likes sticking out on the chain, changing how the molecule partners up in reactions. Dithiocyanate groups give the molecule teeth, letting it link up with metals, form stable complexes, or get involved in the making of larger organic frameworks. Its molecular weight and probable low volatility suggest it is more a sticky liquid or soft solid than a drifting gas, and its solubility in polar versus nonpolar solvents always draws interest for separation work. Scientists who have handled similar molecules know the scent can be sharp and the texture varies, usually indicating a need for fresh air and decent gloves if working at the bench.
One thing that stands out about working with molecules like 2-Dimethylamino-1,3-dithiocyanatopropane is the attention paid to labeling and regulations in any serious laboratory. I’ve sat through enough safety briefings to appreciate up-to-date labeling: clear hazard warnings, date of preparation, responsible researcher. Anyone who’s handled dithiocyanate derivatives knows skin contact or inhalation can spell trouble. The story gets more complicated for international shipments, where packing rules, hazard designations, and transit paperwork change country by country. In a well-run lab, the bottle sits inside a secondary container and only the most up-to-date safety sheet accompanies it. That’s not just bureaucracy—it’s protection for everyone involved, especially when compounds are new or the toxicity hasn’t been perfectly pinned down yet.
Preparing 2-Dimethylamino-1,3-dithiocyanatopropane usually involves a careful multistep synthesis, starting with a dimethylamino-propane precursor. Chemists attach dithiocyanate groups through a staged reaction, often involving thiocyanate salts and halide exchange chemistry in a well-stirred flask. These reactions can generate unpleasant byproducts like hydrogen sulfide or free thioacids, which means working in a hood with solid ventilation isn’t just a recommendation—it’s required. The yield varies by the purity of reagents and the patience of the scientist. Once isolated, the product often needs purification by crystallization or chromatography. Every chemist who’s worked in synthetic labs has tales of delicate distillation assemblies and the ever-annoying clog of sulfur-laden goo, which reminds everyone how the properties of functional groups really matter in bench work.
Chemists love a molecule that does more than sit still, and this one brings plenty of character to the table. That dithiocyanate pair can latch onto certain metals or get swapped out in nucleophilic substitution reactions. The dimethylamino handle gives synthetic flexibility, letting scientists twist the backbone into larger molecules, or convert it into salts or other derivatives. This sort of flexibility spawns direct research uses and also opens paths for modification that matter in industrial and medicinal projects. I’ve seen similar structures used to tweak dyes, add reactivity to ligands, or create intermediates that can head in almost any direction. Every time someone finds a new trick with these functional groups, the downstream effects ripple across more than one field.
Anyone poking through the literature finds that 2-Dimethylamino-1,3-dithiocyanatopropane hides under more than one name. Depending on the catalogue or the researcher, alternate labels can include terms based on IUPAC or even short-hand abbreviations, but the structure draws the most attention for its two thiocyanate groups and the unmistakable dimethylamino appendage. The diversity of names sometimes leads to confusion, especially in older papers or cross-referenced patents. This is where clear communication—between suppliers, researchers, and manufacturers—matters as much as the molecular recipe.
People who spend their careers in chemistry quickly realize that safety protocols aren’t just legal hoops—they’re stitched into every responsible step. Handling a compound like this one, with its sulfur and nitrogen content, means wearing nitrile gloves, keeping goggles on, and checking the label twice before reaching for a pipette. Good ventilation turns from a suggestion into a requirement. Storage in a cool, dry place, away from acids or oxidizing substances, can prevent messy incidents. Lab culture grows around this kind of discipline. More than a few lessons come from mistakes—a cracked bottle, a careless pipetting session, or a forgotten fume hood. These real-life incidents probably inform more safety updates and operational standards than any single textbook.
As the chemical toolbox expands, structures like 2-Dimethylamino-1,3-dithiocyanatopropane keep popping up in research papers, especially in fields looking at ligands for metal complexation, dye intermediates, or precursors for tougher networking polymers. Medicinal chemists sometimes use sulfur- and nitrogen-rich compounds as potential leads in drug development, aiming for selectivity in reactions with biological targets. Some materials scientists tweak similar molecules looking for better adhesion, corrosion resistance, or sensitivity in detection systems. Anyone who’s worked with dyes or sensors knows that small substitutions in the chain can mean big changes in color or activity.
Every new compound starts with curiosity, but research quickly gets practical. Current work on derivatives like this tackles new synthetic pathways, studies stability under heat or UV stress, and explores reactivity with biological systems. There’s real value in teasing apart how the functional groups interact with proteins, DNA, or metal surfaces. Graduate students and postdocs know the frustrations and breakthroughs that come when a planned reaction delivers something unexpected—sometimes sparking a fresh line of inquiry about side reactions or decomposition pathways. Research groups push for detailed characterization, using NMR, IR, and more high-res analysis than older labs ever dreamed of. The depth of these studies supports both academic discovery and industrial advances.
Chemists rank toxicity alongside reactivity whenever a new compound enters the lab. Compounds with dithiocyanate groups grab attention because of concerns about skin absorption, respiratory irritation, and—if enough exposure—the long-term effects on organ function. Early toxicity screens focus on acute effects: burning eyes, skin rash, cough. Longer-term studies rely on animal models or in vitro systems, but gathering reliable data takes time and careful reporting. Many government and regulatory agencies update their guidance slowly, only after enough case reports land on the desk. It falls to lab managers and research directors to keep track of new toxicity findings and update protocols on a rolling basis. This means the burden of safety research falls both on the official reports and the careful documentation of anyone who’s handled the compound in the lab or in manufacturing.
Interest in multi-functional organic molecules keeps growing, especially as industries move toward greener or more efficient solutions. Researchers keep hunting for routes that save steps, cut down hazardous byproducts, and make scale-up less risky. The mix of nitrogen and sulfur in 2-Dimethylamino-1,3-dithiocyanatopropane aligns well with needs for more adaptable ligands, faster reacting intermediates, or even controlled-release structures in pharma and agrochemical sectors. Teams working in computational chemistry model how these compounds could behave in larger, engineered systems, hoping to pick winners before stepping into synthesis. Methods for assessing safety and downstream environmental impact are catching up, integrating new data faster than before. In my years watching innovation cycles, the story rarely ends with the molecule itself—what matters is how teams adapt it, study its quirks, and keep pushing the limits of what it can do.
It's easy to pass over chemicals with names that feel more like a tongue-twister than anything you might encounter in daily life. But 2-Dimethylamino-1,3-dithiocyanatopropane has carved out a useful spot in several fields, particularly where industry and research overlap. Speaking as someone familiar with both chemistry classrooms and busy manufacturing floors, I see this compound come up in conversations about innovation.
Pharmaceutical development doesn’t get far without specialty chemicals like this one. Researchers often use 2-Dimethylamino-1,3-dithiocyanatopropane to kick off reactions for synthesizing more complex molecules. Its dithiocyanate groups give it the kind of reactivity needed for building advanced organic compounds. There’s real value when scientists can count on specific building blocks—this one sits in that category.
For medicines, the compound helps assemble molecules with antiviral, antibacterial, or anticancer properties. That doesn’t mean it shows up in your prescriptions at the pharmacy, but it’s there behind the scenes, helping chemists try out new drug designs. The process of finding novel pharmaceutical candidates gets quicker with reliable reagents like this.
Moving beyond medicine, this molecule shows up in manufacturing specialty dyes. Its reactivity makes it perfect for introducing sulfur and nitrogen groups into dye structures, giving more vibrant colors and better durability. Textile factories and ink producers looking for specific shades or properties sometimes reach for compounds that start with 2-Dimethylamino-1,3-dithiocyanatopropane. Strong, stable colors help keep shirts from fading after just a few washes, and vivid pigments don’t just pop off the page in glossy magazines—they rely on chemistry, too.
The changing world of electronics benefits as well. Modern circuit technology depends on custom molecules to tweak semiconductor properties. Because this compound offers both amine and dithiocyanate functionality, it’s used in the development of new sensor materials and conductive coatings. Over the years, chemists searching for tailored materials that fit specific needs in microchips or solar panels have experimented with it to fine-tune things like charge transfer or light sensitivity.
Of course, working with any powerful reagent comes with responsibility. You can't just toss chemicals like this around without thinking about safety or environmental impact. Proper ventilation, gloves, and training aren’t optional extras—they’re part of daily reality for anyone handling this in a lab or production facility. Chemical companies and academic groups both invest in better handling procedures, waste treatment, and safer reaction conditions to tackle the risks in meaningful ways.
Scaling up from a flask in the lab to a reactor on the plant floor requires real commitment to responsible manufacturing. That means not just watching costs, but also reducing byproducts and keeping emissions down. Best practices, continuous monitoring, and environmental stewardship each play a part in making sure that progress in chemistry doesn’t come at an outsized cost to health or safety.
2-Dimethylamino-1,3-dithiocyanatopropane represents more than just a tool for the toolbox. Its journey from research bench to real-world applications shows how clever design can open up opportunities in health, industry, and technology. The trade-off between innovation and safety stays right at the center of any discussion about the future of specialty chemistry, and that’s a conversation everyone should care about.
Chemistry can look like a world of indecipherable lines and numbers. But under those tangled names live molecules shaping medicine, tech, and daily safety. Take 2-Dimethylamino-1,3-dithiocyanatopropane. Out of context, that sounds like pure jargon, but this molecule tells a subtle story about the importance of knowing exactly what’s in your beaker or bottle.
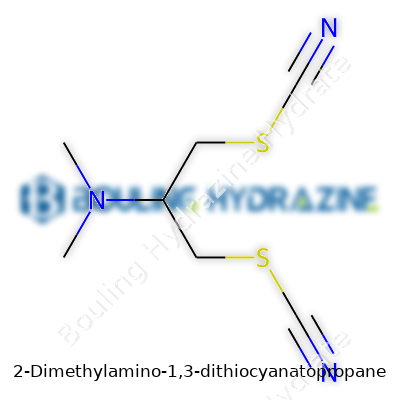
This compound sports two dithiocyanato groups with a dimethylamino side chain hanging off a propane backbone. If you break the name apart, you get a clear sense of its build: three carbons for propane at the center, with a pair of thiocyanate groups (SCN) at positions one and three, and a dimethylamino (N(CH₃)₂) group at position two. Chemically, that builds into the formula C7H11N3S2. The molecular weight lands at about 201.31 g/mol.
Years of fascination with chemistry taught me that a minor tweak to a molecule’s makeup changes almost everything about how it behaves. Add a methyl or swap a sulfur and you’re suddenly looking at a different smell, solubility, or even an entirely new function. With 2-Dimethylamino-1,3-dithiocyanatopropane, those sulfur and nitrogen features often catch the eye of researchers scoping out compounds for further development. Components like thiocyanate stand out in pharmaceutical or agricultural research since the presence of sulfur atoms can spark unique reactivity or biological activity.
There’s good reason for attention to precise structure in any setting where chemical safety, efficacy, or innovation need to be spot-on. Knowing the molecular formula lets chemists calculate not just purity or dosage, but also hazardous potential or breakdown products that come from mishandling.
More than once, I’ve seen labs get stuck—or worse, misled—by missing or incorrect formula data. You don't just trust that what’s on the label matches the compound in practice. Pulled from reputable literature or peer-reviewed reports, accurate chemical formulas stop mistakes before they begin, especially for new researchers who might not catch a typo but could pay for it with failed tests or wasted hours.
Reliable formula data ripples past the scientist’s bench. It directs suppliers, educators, regulators, and emergency workers. In a world flooded with unverified shortcuts and ask-no-questions resellers, transparency and verification separate science from speculation.
The lesson from any complex compound, including 2-Dimethylamino-1,3-dithiocyanatopropane, comes down to rigor and respect for detail. Anyone handling novel chemicals can help move things forward by making structure and properties easy to check and hard to fake. Pushing for open, peer-reviewed databases and routine third-party checks keeps research grounded.
Science thrives on eager minds and precise facts in equal measure. For every hard-to-pronounce molecule, it’s not the syllables that carry weight—it’s the knowledge, accuracy, and trust built from the formula up.
Spending years poking around in labs and industrial environments, I’ve learned that respect for chemicals starts with preparation, not panic. 2-Dimethylamino-1,3-dithiocyanatopropane belongs to a group of compounds that don’t give second chances to those cutting corners. This isn’t about scaring anyone away from science. It’s about facing facts: some chemicals pack enough risk to make rules matter.
Gloves made of nitrile or neoprene, safety goggles with side shields, and a fitted lab coat with wrist closures become as important as your pipettes when using something like this. Skin absorption can become a problem fast with dithiocyanate compounds. Splash risks, fumes, and stains on your hands can turn into something worse than a ruined weekend. Your eyes matter more than multitasking—chemical splash goggles should never gather dust at the back of your drawer.
Compounds bearing strong odors often give a warning before trouble, but faint or nearly scentless ones can leave you guessing. Relying on a well-maintained fume hood isn’t fancy, it’s basic lab living. Engineering controls—like local exhaust fans and well-designed workspace layouts—limit exposures. Never trust open windows or the promise of “just for a minute.”
Spills never ask for permission. Years back, a quick search for a misplaced beaker taught me that clutter can become costly—think cross-contamination, accidental mixing, or heat sources meeting volatile chemicals. Verbal reminders and checklists don’t just sound official; they keep projects moving smoothly with fewer headaches. Double-checking labels, dating containers, and storing reagents according to their compatibility turns an often-ignored step into a lifesaver.
Small spills can spiral into emergencies. Absorbent pads and proper disposal bins belong right next to your bench. Too many treat cleanup as an afterthought. Chlorinated solvents or flammable liquids should never go down the drain, and dithiocyanate compounds can pose extra hazards, depending on what they contact. Review safe disposal procedures before, not after, any experiments. Corporate labs and home tinkerers both benefit from clear protocols.
Anyone handling reactive organic compounds should know the usual warning signs of trouble: skin irritation, shortness of breath, headaches, or unexpected rashes. Don’t wait for something “serious.” Report accidents, seek immediate care, and follow up with a health professional. Too many people try to shrug things off at their own cost.
Annual safety training may sound repetitive, yet every single case of lab injury I’ve seen involved someone sure they “knew better.” Practicing with mock spills and reviewing up-to-date handling practices pays off. Online trainings, quick reads of the latest chemical data sheets, and honest chats with more experienced colleagues build habits that last.
If you work with complicated chemicals like 2-Dimethylamino-1,3-dithiocyanatopropane, your actions ripple out to everyone nearby. Clean hands, tidy benches, and a working knowledge of first aid matter just as much as successful reactions. The science gets better, and so do the odds of making it home healthy after every shift.
Anyone who’s worked with specialty chemicals like 2-Dimethylamino-1,3-dithiocyanatopropane knows the fuss around storage isn’t only paperwork. Manufacturers write up elaborate protocol for a reason: it’s about protecting people and accuracy in research or industrial applications. Ignoring proper storage shaves more than just shelf-life; it can introduce dangerous byproducts or cause the material to lose its punch. If you’ve opened an old bottle to find a whiff of rotten eggs or a lump instead of a powder, you know shortcuts are a trap.
This compound’s thio and amine groups grab moisture and react with oxygen pretty readily. Too much humidity, and you risk caking, decomposition, or worse yet—dangerous off-gassing. Store it where the air stays dry. I once saw a lab lose thousands of dollars in reagents from a “climate-controlled” closet that nobody realized wasn’t keeping up. Even the best container can’t do the job if the air is sticky.
Sunlight and high temperatures break down sensitive chemicals fast. Light especially can spur unwanted reactions in sulfur-based compounds, so keep it in darkness. Ordinary shelf space just doesn’t cut it for materials that break down under everyday lab light. Use amber glass bottles, and stash them away from direct rays—even a lab window during winter can mess up your inventory without any warning signs.
Not every plastic or metal can handle thiocyanate chemistry. A glass container, lined with a PTFE-sealed cap, blocks air and keeps the environment stable. Some plastics, if you're unlucky, actually react with the chemical, causing slow leaks you can’t see until it’s too late.
Cold storage keeps reactions to a minimum. Refrigerators set around 4°C work for many labs, but a freezer adds extra insurance for long-term stashing. Don’t over-pack freezers, though; opening and closing introduces condensation. If you’re tight on space, dedicate a smaller fridge, label everything, and stick to scheduled checks.
A locked chemical storage cabinet reduces access and minimizes the chances that an untrained hand grabs the bottle. Always use clear hazard labels, not just chemical names, since mistakes happen. Adequate training matters even more than a fancy storage space. Run short refreshers every few months so everyone remembers why these precautions exist.
Accidental contact or inhalation risks push up the stakes. Spill containment trays underneath every bottle, along with spill kits nearby, prevent a little slip-up from turning into a disaster. Simple habits—gloves on, lids tight, goggles down—pay off in the long run.
People in labs sometimes treat storage guidelines as optional, but poorly-stored chemicals sabotage both the science and the safety. Reliable protocols reduce waste, improve experiment results, and avoid emergencies—in other words, they make everyone’s job easier and safer. Safe chemical storage isn’t just a technical point; it means supporting everyone in the workplace.
Working with hazardous chemicals sharpens a sense of caution, especially with ones like 2-dimethylamino-1,3-dithiocyanatopropane. In the lab, this compound keeps everyone respectful. Anyone who has even gotten a whiff knows it’s no joke—sulfurous, bitter, and sticks to your clothing and skin.
Risk isn’t a line in a textbook. It seeps into your daily routine: gloves, goggles, splash apron, and always a fit-tested fume hood. These chemicals can cause real damage—eye irritation, skin burns, or worse. The disposal question isn't just a regulatory issue; it’s about keeping everyone on your team safe. I’ve seen fingers blister and lungs burned by shortcuts that traded speed for safety.
Down the drain is not an option. Many chemicals, especially organosulfur compounds, run toxic both to people and to aquatic life. Wastewater treatment can’t keep up with every exotic reagent, especially things designed never to biodegrade in the first place.
Improper disposal can lead to contamination of groundwater and soil. In the United States, the Environmental Protection Agency’s Resource Conservation and Recovery Act (RCRA) puts a strong fence around hazardous waste. Local landfills reject this type of chemical for good reason—the risk doesn’t stop at the lab’s exit.
In practical terms, the safest route involves the professionals. University and industrial labs place this waste in clearly labeled, tight-sealing containers. These containers go into secure storage until hazardous waste management staff collect them. This approach limits exposure, and the paperwork tracks the chemical from cradle to grave.
Incineration at permitted facilities offers the most trusted solution. High-temperature, controlled oxidation destroys organic chemicals and keeps sulfur byproducts contained in scrubbers, instead of letting them drift out with the wind. This process keeps the nasty stuff out of waterways, off the ground, and out of the food chain.
Shortcuts have a way of coming back around. A friend of mine found out the hard way after a sloppy disposal led to an evacuation and regulatory investigation. Lives get upended in these situations—fines, lawsuits, damaged reputations. There’s a reason why trained chemical hygiene officers check and double-check waste protocols.
Some labs try to neutralize or treat waste in-house. That’s risky ground. Without exact procedures—often closely held and updated by regulatory agencies—you run the risk of creating something even more dangerous, like toxic gases or unstable byproducts. Handling 2-dimethylamino-1,3-dithiocyanatopropane is not a hobbyist’s project.
Proper disposal starts with training and clear rules. New lab members don’t just read policies—they watch experienced hands handle waste, and veterans answer their questions. Trust builds around habits that put safety first, every single time.
A chemical’s story shouldn’t end with a disaster in the headlines. Every step, from ordering to disposal, invites personal buy-in. Respecting the science means trusting the process—not reinventing it on a whim. Following the recommended path for 2-dimethylamino-1,3-dithiocyanatopropane disposal isn’t just legal; it’s the anchor that keeps the whole operation upright, safe, and sustainable.