2,3,4,4'-Tetrahydroxybenzophenone draws a curious crowd in the world of chemical research. Decades ago, it popped up in synthetic chemistry studies as scientists looked for better ways to protect polymers and increase product longevity. History doesn’t unfold with fanfare for compounds like this, but as demands for stability in plastics and resins shot up, chemists found more practical use for this benzophenone derivative. The road to its common use began in polymer science labs, where its simple structure attracted attention for both experimentation and commercial application.
Looking at 2,3,4,4'-Tetrahydroxybenzophenone, the raw facts don’t do much until you recognize its impact in making polymers and coatings last longer. What sits in a glass vial as an off-white powder ends up helping plastics survive sunlight and years of wear. In real-world terms, it holds up as a UV absorber, fighting off the harshness of sunlight and stopping those cracked garden chairs or faded dashboards. You don’t need an advanced degree to grasp that most folks, myself included, benefit from things that last. Products that can make materials last longer become less of a chemical curiosity and more of a quiet workhorse behind everyday goods.
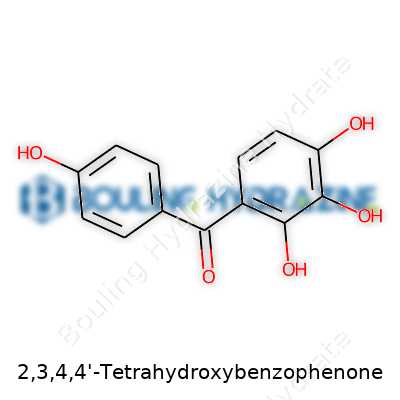
After you’ve seen enough chemicals, you start paying attention to what a structure really brings to the table. The benzophenone core, dotted with four hydroxyl groups, tells you this isn’t a simple molecule. Its structure gives it specific reactivity and a knack for forming hydrogen bonds, which matters when you push it into a polymer blend. Color and taste aren’t of much use here, but a high melting point and moderate solubility in organic solvents make it manageable in most lab and industrial settings. The real key is how it absorbs ultraviolet light; its chemical backbone means it soaks up rays at just the right wavelength to protect plastics and coatings from UV-induced breakdown.
Nobody wants a product mislabeled or misused, especially not when handling chemicals. Regulatory bodies push for clear identification—including proper synonyms, purity standards, and batch testing—so accidents get avoided. Standard packaging and clear hazard labels do more than satisfy paperwork; they guard against slip-ups. I remember spending long hours in the lab sorting through chemical stock and missing or vague labels made everyone jumpy, especially with powders like these that look like so many others. Exact specs depend on intended use, but purity, moisture content, and absence of impurities set benchmarks for each application.
The chemical world rarely prizes brute force. Making 2,3,4,4'-Tetrahydroxybenzophenone involves multi-step synthesis, often beginning with the right benzene derivatives and using controlled oxidation or hydrolysis. In practice, experienced chemists fuss over reaction conditions—temperature, catalyst choice, solvents—because a missed beat can tank the yield or introduce unwanted byproducts. Careful solvent extraction and crystallization separate out the product, but scaling up always brings a different set of headaches. Folks in R&D sweat these steps so end users get a consistent, effective chemical, whether it heads to an academic lab, factory floor, or a refinement mill.
Tetrahydroxybenzophenones don’t rest idle; their structure invites functional tweaking. The hydroxyl groups make it possible to introduce further modifications through alkylation, esterification, or ether formation. Tinkering like this fine-tunes how the compound interacts with target polymers or biological systems. On the benchtop, watching these transformations plays out as a lesson in how chemistry adapts to meet demand. For example, making a more lipophilic derivative suits certain resin applications where water resistance trumps other factors. If you ever tried to tailor a polymer’s performance, you’d appreciate the flexibility built into this molecule.
Naming in chemistry often feels like a walk through a maze. 2,3,4,4'-Tetrahydroxybenzophenone goes by several names, including its systematic title and a few commercial monikers. Synonyms can cause confusion, especially across borders or industries. Anyone who’s worked on international collaborations knows the pain of cross-referencing chemical registries. The drive toward unique identifiers—like CAS numbers—has helped, but users still lean heavily on familiar trade names and shortcut labels. Recognizing the diverse aliases for a compound is more than a paperwork issue; it minimizes miscommunication and mistakes.
Workplace safety isn’t optional—too many ignored warnings have cost lives. Handling tetrahydroxybenzophenone means respecting its potential hazards: inhalation risk, skin contact, possible dust explosions, and environmental damage. Operators use gloves, eye protection, and well-ventilated spaces as a matter of routine. Safety data sheets give guidance, but they can’t replace hands-on training. I’ve seen new technicians handle powders with an odd mix of caution and overconfidence, and it doesn’t take much to create problems—skin irritation, unintended reactions with nearby chemicals, or spills that escalate quickly if ignored. Regular safety audits and clear signage keep standards up, making sure one oversight doesn’t take out half a lab.
The molecule’s real worth shows in what it makes possible. In plastics, 2,3,4,4'-Tetrahydroxybenzophenone shields against UV, holding together picnic plates, auto parts, and electronic housings after years in harsh sun. Textile makers see benefits when colors stay bright and fibers resist yellowing. In coatings and paints, it steps between harsh sunlight and substrate. Research continues to look for new uses—cosmetic formulators experiment with it as a sun-blocking agent in special products, although safety limits usually steer them toward other ingredients. Each industry finds a niche, and the common thread is extending the life and visual appeal of countless goods.
Behind commercial success hides years of testing and frustration. R&D teams keep pushing to give this compound a broader, safer range. Some explore blending it with newer polymers or seeking derivatives with lower toxicity or better solubility. Others try to synthesize it more efficiently or look for applications outside plastics, such as pharmaceuticals or photovoltaic cells. The process isn’t all lab coats and perfect results. Many leads fizzle, and the rare breakthrough drives the field forward. I’ve watched teams dig into minute structural tweaks hoping for a big performance leap, reminding everyone innovation means risk plus persistence.
Interest in environmental and human health has cast a harsher light on compounds like 2,3,4,4'-Tetrahydroxybenzophenone. Studies have flagged both potential cytotoxic effects and broader ecological risks. Biodegradability, bioaccumulation, and aquatic toxicity all challenge widespread use, as do evolving regulations around persistent organic pollutants. Scientists dive into animal models and cell cultures, hoping to untangle side effects and chronic exposure impacts. Not all data paints a clear picture—much depends on dose, route, and exposure scenario. Calls for more testing echo through journals and oversight committees alike; nobody wants a convenient additive turning into tomorrow’s environmental problem.
The next act for 2,3,4,4'-Tetrahydroxybenzophenone will depend on balancing performance with responsibility. Better synthesis routes could slash waste and energy demand. Greener derivatives might address mounting toxicity concerns. Continued research could unlock new uses, especially as industries look for cost-effective, sustainable options that don’t fall foul of tightening laws. It’s a case of learning from the past—where overlooked side effects of chemical progress spawned new challenges—and seeking innovation that puts safety and durability on equal footing. As someone who values both the long life of practical goods and a clean bill of health for people and planet, I see persistent effort in these areas as the only sensible path forward.
In the world of specialty chemicals, most people outside the lab never hear about compounds like 2,3,4,4'-Tetrahydroxybenzophenone. Yet, for plenty of products that cross our paths each day, this colorless powder quietly does its job. Most often, you’ll find it as a stabilizer in adhesives, coatings, and plastics. Chemically, it blocks harsh ultraviolet (UV) rays, and that small bit of magic ends up playing a pretty big role in keeping materials strong and looking good.
Growing up in a neighborhood filled with summer sun, I watched countless lawn chairs and plastic patio tables warp or fade in just a season or two. At the time, it seemed like magic when some brands stayed looking sharp after whole summers outside. The trick? Stronger UV protection. Additives like 2,3,4,4'-Tetrahydroxybenzophenone absorb that harsh sunlight and protect both the plastic’s surface and whatever’s inside or on top.
We see this effect in coatings, too. Think about that shiny finish on electronics, car panels, and building materials. UV rays cause surfaces to chalk, peel, or shift colors. By mixing in strong UV blockers, manufacturers delay this breakdown. Products last longer and hold their value. Rather than spending money to replace faded and weakened outdoor goods every year, consumers get mileage out of what they buy.
Safety sometimes enters the discussion with specialty chemicals. 2,3,4,4'-Tetrahydroxybenzophenone isn’t a known troublemaker for the environment or human health, but as with most chemical compounds, handling deserves care. Responsible producers stick to strict regulations during manufacture and disposal, not because it’s flashy but because it’s smart for workers, consumers, and the planet. Labs continue monitoring these chemicals for any evidence of harm, following global standards set by governments and organizations with deep expertise, such as the Environmental Protection Agency and health departments.
Nobody thinks about chemical additives when pulling leftovers from a plastic container or unwrapping a new phone. Yet, smart chemistry helps products stand up to heat, light, and time. The path to stronger, longer-lasting goods won’t end with what worked yesterday. Ongoing research can improve how 2,3,4,4'-Tetrahydroxybenzophenone functions—maybe making it more efficient at blocking UV or easier to recycle after use. For industries focused on performance and sustainability, there’s room to push past the status quo.
I’ve seen companies rethink their formulas to find safer, greener materials, especially as public interest in eco-friendly products grows. Sometimes, the answer isn’t swapping out an ingredient, but finding clever ways to use less of it or make sure it doesn’t wind up in a landfill. By listening to chemists, consumers, and regulators, industries can steer toward products that keep both people and the environment in mind.
You probably won’t spot 2,3,4,4'-Tetrahydroxybenzophenone on any shelf, but its behind-the-scenes work shows up in the durability and longevity of everyday items. Pushing for better safety and responsible use with every batch not only meets strict regulations but matches what most of us want out of modern life: less waste, more value, and products built to last.
Sunscreens, plastics, hair sprays, and even paints. Chemicals like 2,3,4,4'-Tetrahydroxybenzophenone pop up in all sorts of everyday products, and many folks may not recognize the names printed in fine print. This compound goes in because it absorbs ultraviolet (UV) light, helping products withstand sun exposure and sometimes preserving color and structure. What seems like a technical convenience, though, leads to a closer look—something that concerns anyone interested in health and long-term safety.
Science should always guide us, but results don’t always land clearly. Toxicology testing on similar benzophenones shows these compounds can absorb through the skin and sometimes disrupt hormones in animal studies. Researchers noticed some effects on reproductive hormones and thyroid functions in animals—less so in humans, but regulators pay attention whenever endocrine activity comes up. At moderate to high doses, certain benzophenone derivatives (not exactly this one, but cousins in the same chemical family) raised red flags for long-term exposure and potential links to hormone activity in large animal studies.
When looking for human studies, the landscape thins out. Available research focuses on close relatives rather than this exact form, though chemical structure suggests similarities that matter. Europe handles substances like these with strict rules; a few countries restrict overall use of several benzophenone-type UV filters, especially in kids’ products. In the United States, the FDA maintains a list of allowed sunscreen ingredients, but currently this particular derivative does not receive the same level of everyday scrutiny as well-known others like oxybenzone.
As a parent, I scan ingredient lists out of habit, realizing I just want to answer, “Will this bother my kids? Will it stick around in our bodies?” Compared to other substances, clear answers on 2,3,4,4'-Tetrahydroxybenzophenone safety still feel out of reach, especially for long-term or high-frequency use. I trust my local pharmacist with questions but usually end up double-checking scientific sites or published studies—hoping to cut through jargon. Nothing replaces good regulation and transparency. Limited data about this chemical doesn’t mean it’s trouble, but it means people can’t make completely informed choices.
Product manufacturers win points for disclosing full ingredient lists and supporting outside testing. Regulators earn trust when they update policies to match current research, even if that means stepping up scrutiny on chemicals where human health studies lag behind. Doctors and scientists need public funding to track these chemicals in real, everyday populations—not just in labs with high doses and isolated cells. Parents and consumers can press for these changes, asking companies and lawmakers to take these steps seriously instead of leaving all responsibility with buyers.
There’s also room for simple fixes. Buying products labeled for minimal or no UV filters (when possible), trying alternatives with proven safety history, and calling companies to account for vague labeling each help. No one wants to trade UV protection for uncertainty over hormone disruption, and informed choice starts with knowing what chemicals do inside and outside the lab.
Calling for more research and stronger regulations doesn’t mean stopping progress. It shields the public when science hasn’t closed the book on long-term safety. Chemicals like 2,3,4,4'-Tetrahydroxybenzophenone bring benefits, but trust follows clear evidence, transparency, and a willingness to adapt policies as more comes to light.
It’s pretty rare to hear someone at a coffee shop chatting about tetrahydroxybenzophenone. This molecule, known more formally as 2,3,4,4’-tetrahydroxybenzophenone, might not sound familiar, but dig a little and you’ll find it in the guts of real-world chemistry. I once stumbled on it while helping a friend research safer additives for plastics. When you open up the books, tetrahydroxybenzophenone sits in the benzophenone family, loaded with four hydroxy groups and a core built on two aromatic rings separated by a carbonyl group.
You put a pure sample of this compound on the benchtop and usually spot a crystalline, off-white powder. It doesn’t have much of an odor, so this one won’t announce its presence across the room. The molecule packs in four hydroxyl groups, which pull in water molecules — so it tends to absorb moisture from the air given the chance. For practical handling, that hygroscopic nature often sends chemists reaching for gloves and sealed containers.
In my grad school days, compounds like this signaled “phenol territory.” With hydroxyl groups across the rings, tetrahydroxybenzophenone behaves as a polyphenol. It holds its own as a mild acid, more so than its non-hydroxy cousins, since those OH groups happily donate protons. In reactions, this makes it a handy candidate for catching free radicals—a key trait for anyone concerned about plastics degrading under sunlight. Those same hydroxyls love to form hydrogen bonds, so tetrahydroxybenzophenone hangs tight with other polar compounds and mixes smoothly in solvents like ethanol or acetone.
Run it through a UV spectrophotometer and you’ll see it gobbles up ultraviolet light. This trait pops up in sunscreen chemistry, often with relatives of benzophenone. In the world of polymers, tossing in tetrahydroxybenzophenone helps protect products from the relentless beatdown of sunlight, slowing down the yellowing and brittleness that comes with age.
Some might think of specialty molecules as a niche pursuit, but these properties ripple outward. In plastics and coatings, manufacturers face a big headache: how to extend product lifespans in outdoor and industrial environments. Compounds like tetrahydroxybenzophenone serve as armor, locking up reactive oxygen species and helping materials “live” longer. My experience with product teams has taught me that reliable additives don’t just mean fewer recalls—they translate to less waste, reduced costs, and more trust in daily products.
Most chemists recognize that with strong antioxidants often comes a question about toxicity and environmental persistence. Tetrahydroxybenzophenone, no surprise, can cause issues if mishandled. Research continues on its safety in consumer goods. Studies point out that extended skin contact or leaching from plastics should be limited. The better labs keep finding less persistent, safer analogs with similar performance.
What can manufacturers do? Staying open to green chemistry tools, like lifecycle analysis, pushes companies to compare these stabilizers against biodegradable alternatives. Policies worldwide keep tightening, so switching to solutions that break down after use—without health risks—seems smart in the long run.
Looking past the tough name, tetrahydroxybenzophenone matters to anyone who wants the things they own to last under the sun. Paying attention to its chemistry gives us the power to demand safer, smarter products down the road.
Working with chemicals like 2,3,4,4'-Tetrahydroxybenzophenone introduces more than tough pronunciation. This compound comes up often in labs and the chemical industry, showing up in some plastics and coatings. Proper storage rarely gets discussed in plain language, but mistakes can create real hazards for people and property.
At school, I watched a classmate ignore labeling rules for a similar chemical. That carelessness almost kicked off a fire when incompatible solvents mixed. Stories like that stick with me and make chemical storage a lot more personal. Nearly every serious chemical accident I’ve read about starts with small lapses—unlabeled containers, lids left open, random shelf spots.
2,3,4,4'-Tetrahydroxybenzophenone sits in a class of organic compounds that may degrade if exposed to light, air, or moisture. The key is creating an environment that slows down these processes—dry containers, tight seals, and storage away from sunlight. Moisture turns many powders into sticky clumps or even triggers dangerous breakdown. UV light from a window or overhead lamp can speed up reactions, sometimes generating hazardous byproducts.
A lot of people only worry about explosion risks. The reality is, poorly stored chemicals often end up as waste or leaks—straight into trash, drains, or the air. 2,3,4,4'-Tetrahydroxybenzophenone contaminating water can impact local wildlife or even reach the food chain. Simple routines like double-checking seals and using airtight amber glass bottles make a bigger difference than the latest high-tech gadget.
OSHA and the CDC hammer home that labeling and containment control spill risks and costly cleanups. Insurance companies don’t care about abstract rules—they want to avoid real payouts on accidents, which start with bad storage habits. An overlooked chemical can outgas or change its own structure, creating invisible risks over time.
In practice, the smartest move is maintaining a dry, cool spot—out of reach of sunlight and humidity. I’ve seen custodians and researchers label storage cabinets by chemical class, so users can avoid cross-mistakes. Silica gel packets squeezed inside containers suck up sneaky moisture. Keeping chemicals in their original packaging knocks out confusion about expiration or composition, especially after months on the shelf.
Most labs rely on chemical inventory systems that track storage dates and remind staff about old stock. Always store 2,3,4,4'-Tetrahydroxybenzophenone away from strong acids, bases, or anything flammable. If a researcher has a question, a senior chemist or safety officer usually knows the hard-earned tricks for that facility.
No law or regulation replaces common sense. If you handle a compound in your work or studies, treat storage like a core skill instead of an afterthought. Lazy habits invite accidents, but storing chemicals the right way takes minutes and pays off in peace of mind. Good storage doesn’t look impressive—just neat shelves, labeled bottles, and a quiet lab that doesn’t make headlines for the wrong reasons.
Every few years, an obscure-sounding compound moves from lab benches and journal articles to occupy a sudden spotlight as researchers or manufacturers hunt for sources. 2,3,4,4'-Tetrahydroxybenzophenone fits that category. On its face, the name hardly rolls off the tongue, but for chemists, its value is clear. Whether working in academic research, specialty polymers, or materials science, someone eventually needs this compound in pure, verified form. It’s not as simple as dropping by the local chemical outlet, either.
Scrolling through a general chemical supplier catalog doesn’t often present this kind of specialty benzophenone. The big players like Sigma-Aldrich, Alfa Aesar, TCI, and Fisher Scientific make up the first stops for many researchers. When I needed specialty phenolic compounds for polymer crosslinking work, I learned that anything with specific substitution patterns rarely sits waiting on a shelf. If a compound isn't in their database or costs an order of magnitude more than budgeted, disappointment is real. This search teaches patience—sometimes a synthesis feels simpler than navigating the bureaucratic trails and price quotes.
Companies with production capabilities in fine chemicals—think of suppliers specializing in custom or low-volume synthesis—provide one legitimate avenue. The downside: minimum order quantities and setup fees quickly balloon, making this route only sensible for group purchases or well-funded projects. Beyond price, established suppliers have a track record for documentation, purity, and proper handling procedures, which weighs heavily when the wrong impurity could compromise research or endanger safety.
Research chemicals like 2,3,4,4'-Tetrahydroxybenzophenone often fall under various national and international regulations. Before purchase, confirming intended use stays within legal lines matters. Import permits, end-user statements, and institutional oversight often form a gauntlet one can't sidestep. Researchers in academic labs can sometimes piggyback on university bulk orders to simplify things, but individuals and smaller outfits face more hoops.
Online 'marketplaces' offer quick fixes, but risks multiply. Counterfeit or mislabeled chemicals circulate, and sellers willing to ship rare reagents with no questions asked usually offer little recourse if things go wrong. In my lab years, a shipment of so-called “high-purity” material from an online vendor set back weeks of work; the real substance contained a mess of colored byproducts and—more worryingly—a stash of heavy metals. So I only trust certified vendors, no matter the price tag.
Sourcing may seem only a question of who sells what, but behind every bottle stand bigger issues: safety, ethical use, and environmental responsibility. Trustworthy suppliers go beyond paperwork by transparently reporting test results, hazardous ratings, and keeping up with current REACH or TSCA rules. Colleagues in the field echo: saving some cash never justifies sketchy sources. Many chemicals—especially polyphenols—pose substantial health risks in untrained hands, and regulation exists for solid reasons.
There's a growing need for more open infrastructure in sharing rare chemical stocks. Cooperative purchasing networks, run by professional societies or academic consortia, promise to make legitimate access easier. Clearer public databases of legitimate vendors—as seen in Japan’s JST or Europe’s substance registries—shine light on what’s possible. Institutions could support trainees in learning supply chain due diligence through interdisciplinary coursework or mentorship, strengthening both research integrity and personal safety.