People who dig through the history of polymers and specialty chemicals usually stumble onto quite a few names that might not ring any bells but deserve their spotlight. One of those under-the-radar architects is 1,1,1-Tris(4-hydroxyphenyl)ethane—often called THPE in labs. Its story echoes the need for more robust and reliable materials in the evolution of the chemical industry, especially at the point when phenolic resins and high-performance plastics started reshaping engineering, aviation, and electronics. Companies and academic teams took interest in the mid-20th century, when efforts ramped up to synthesize building blocks for resins that could handle heat, wear, and aggressive chemicals. None of that would have moved ahead quite the same without digging into compounds with more intricate molecular layouts. As researchers hunted for ways to improve material properties, THPE got attention for its three-pronged structure and strong phenolic groups—both useful when aiming to reinforce networks in thermosetting polymers and other synthetic materials.
1,1,1-Tris(4-hydroxyphenyl)ethane stands out because it doesn’t do things halfway. With three hydroxyphenyl groups snugged onto a single ethane center, it produces polymers that take punishment where others fall apart. Most people outside chemistry don’t hear about it, but it pops up at the heart of specialty resins, adhesives, and coatings built for the long haul. Its structure lets it form multiple cross-links, giving polymers the strength and resilience you need for electronics housings, structural fibers, and composite panels. It isn’t something you buy over the counter; rather, it gets shipped to manufacturing plants that know how to use its unique chemistry to full advantage. The market is somewhat niche, though, relying on the constant demand for performance that pushes past what commodity plastics can achieve.
THPE doesn’t melt as easily as some basic monomers. Its melting point ticks higher than what you see in standard phenols, which hints at tougher intermolecular forces at work. It comes as a pale, crystalline powder—easy enough to spot in its pure form—and its three phenol groups mean it’s pretty reactive toward some chemical changes. Stir it into the right mix and it stands up to acids, bases, and even decent heat, especially after cross-linking. Unlike simple phenol, THPE offers a broader grid when forming bonds, lending strength and rigidity that makes a difference in high-stress settings. Its solubility leans toward organic solvents that break up hydrogen bonds. THPE also resists absorbing water, which sets it apart from some other resin hardeners and gives a boost to long-term durability in challenging spots.
Chemists working with THPE tend to keep an eye on its purity, since common impurities throw off polymerization. They expect a product with high assay values—upwards of 98 percent—checked by melting point, HPLC, or NMR. Storage also gets extra care, since tightly sealed drums keep out moisture and UV light that could tinker with stability. Regulatory labels highlight the risks of phenolic compounds (skin and eye irritation get a mention), while weighing out sample needs gloves and preferably fume hoods. Batch numbers and standard chemical identifiers stay in line with current best practices for traceability, given the tight standards in electronics and high-performance plastics.
Synthesizing THPE takes planning around multi-step acid catalysis. Most routes kick off with phenol reacting with acetaldehyde, under an acid catalyst to activate and steer the reaction toward a tri-substituted final product. It’s a dance between reaction temperature, catalyst strength, and water control. If moisture slips in, side reactions bump up, lowering the output yield and triggering more purification work. Crystallization steps pull THPE out of reaction broth, followed by repeated washes with solvents to get it pure. Scalability matters because commercial plants can’t get bogged down with low-yield syntheses. People working on research and development have tinkered with catalysts and solvent systems, chasing after greener and safer ways to boost efficiency and shrink waste.
The utility of THPE comes through in the versatility of those three phenol groups. Each group can participate in esterification, etherification, or form strong covalent bonds on polymer chains. That means you get a molecule capable of branching interactions, whether in epoxy adhesives, polycarbonates, or specialty flame-retardant resins. Cross-linking THPE into a polymer increases thermal stability and chemical resistance, putting it at the center of composite build-outs and high-strength thermosets. Sometimes, chemists tweak those phenolic groups further, attaching other functional groups to tailor compatibility with new polymers or enhance reactivity in emerging resin systems. That kind of flexible platform keeps THPE in the running even as technology shifts.
This compound shows up in the literature with a few aliases, enough to trip up even seasoned researchers. THPE remains the most recognized shorthand. You’ll also spot 1,1,1-tri-p-hydroxyphenylethane, 4,4',4''-Ethylidynetrisphenol, and a range of translations from other languages. Sometimes suppliers use custom trade names, yet the structure always revolves around three hydroxy groups aligned on a central ethane carbon. Trademarked blends or modified grades might tweak the branding, especially in the realm of high-tech composites, but at the core it’s the same building block across most catalogues.
Working with THPE in any industrial or research setting takes real safeguards. Phenolic compounds can cause allergic reactions and irritation, so gloves and eye protection rank as standard-issue. Dust control helps reduce inhalation risk. Research journals mention the compound causing mild skin and respiratory irritation if handled irresponsibly, but controlled environments, chemical-resistant gloves, and proper ventilation keep things safe. Regulations call for labeling containers to flag hazards, and waste from synthesis or application must pass through proper neutralization or incineration with trained personnel. Training lab staff, posting clear procedures, and keeping first aid solutions within arm’s reach remains part of the routine.
THPE’s strongest suit rests in specialty polymers—areas where regular plastics give way. Its three-armed structure lets engineers build stronger epoxy adhesives used for mounting electronic components, encapsulating microchips, and even laminating tough printed circuit boards. Manufacturers lean on it to get high glass transition temperatures—meaning finished products hold up in hot environments where normal resins soften or fail. Aerospace components show up on the list, with THPE-enhanced resins finding their way into radomes and interior panels. Its chemical backbone resists breaking down from acids, alkalis, and heat, so performance coatings, specialty paints, electrical insulators, and toughened plastics all benefit. Its triphenol skeleton also gets attention in high-performance polycarbonate blends, which demand clear, stable, flame-resistant parts for lighting, safety glass, and medical equipment. As telecommunication hardware and renewable energy systems keep growing, demand for precision, reliability, and resistance to environmental stress keeps THPE and products like it in play.
Current research doesn’t coast on the successes of the past—they’re looking for ways to lower the environmental impact of THPE synthesis and reduce hazardous byproducts. Green chemistry trends encourage using plant-derived phenol equivalents and milder catalysts. Some teams have targeted new reaction pathways that limit waste and energy use, banking on microwave-assisted synthesis or solid-state catalysis. Another push comes from blending THPE into biodegradable or recyclable resins, in hopes of matching its toughness with a lower waste footprint. Industry-academic partnerships chase these advances, recognizing the need for better environmental credentials as regulations tighten. Clinical and toxicological studies assess its effects on human health, and so far show that with proper handling, risks remain manageable—though new regulations on microplastics and environmental persistence could set the course for further reformulation.
Toxicity always comes up in assessment, especially as compounds with multiple phenol groups evoke caution from health authorities. Acute exposures suggest THPE isn’t as hazardous as many industrial toxins, but skin and eye irritation appear after direct contact. Chronic exposure data remain limited, so ongoing workplace monitoring and medical feedback keep potential risks under scrutiny. Environmental persistence is another area of caution—breakdown rates in soil and water lag behind those typical for mono-phenolic compounds. Testing continues to clarify bioaccumulation risk, keeping regulatory agencies keyed into both production and disposal routes.
Looking ahead, the landscape for THPE could shift along with broader changes in polymer chemistry and manufacturing. Stricter environmental laws, demand for recyclable materials, and consumer pressure could open new avenues for more sustainable production methods. Some researchers already see a future for THPE-modified resins in high-strength, low-weight composite materials needed for wind turbine blades, electric vehicle components, and next-generation medical devices. At the same time, global attention on microplastics signals a need for deeper study on long-term environmental impact. Pushing academic and industrial R&D toward alternatives, safer synthesis, and effective recycling strategies will prove essential. In all this, the qualities that made THPE indispensable—its unmatched strength online with reliable reactivity—will keep motivating chemists to optimize and reinvent the way it’s made and put to use.
Most people haven’t heard of 1,1,1-Tris(4-hydroxyphenyl)ethane, but it fills a quiet gap in manufacturing. Across plastics factories and high-performance material labs, this substance makes waves far beyond its name. I’ve watched chemists blend it like a chef seasons a stew, aiming for strong, resilient results in the things we touch every day—gadgets, home appliances, even medical devices.
The real story comes from its backbone: this molecule’s three hydroxyphenyl arms connect to an ethane core. This structure offers more than just stability. It hands engineers an opportunity to tweak plastics that won’t snap under pressure and can handle heat, moisture, or chemicals. I’ve asked coworkers in resin production what makes top-tier circuit boards last longer, and their answer often points to these hidden molecules.
A lot of focus sits on polycarbonates, those clear, tough plastics in glasses lenses and the covers protecting outdoor lights. The secret lies in the additives that stop the material from yellowing or cracking with time. Blending in 1,1,1-Tris(4-hydroxyphenyl)ethane raises those qualities. This directly benefits anyone using gadgets under the sun, like car dashboards or electronics built for the long haul. An improvement I’ve seen in my own phone case—a little more toughness, less worry about dropping it.
Branching into epoxy resins, this compound brings strength where it counts. Think about wind turbine blades, coatings on bridges, hospital equipment. Engineers want materials that won’t let them down after years of use. A stronger network of chemical bonds, thanks to this additive, keeps these applications running longer. Less maintenance means less waste—something more people care about now, with landfills and pollution in the headlines.
No one wants to swap quality for safety. Not every strong additive stays in use; some come under heavy scrutiny for health reasons, both for workers and end users. Safety authorities test these building blocks and keep a close eye on how companies manage them. So far, data around 1,1,1-Tris(4-hydroxyphenyl)ethane doesn’t ring the alarm bells that certain flame retardants and plasticizers have. Still, I’ve seen calls in journals for more testing, especially as demand grows. Consumer trust stands or falls on transparency.
Building greener processes is another challenge. Factories now sort their chemicals more efficiently and recycle waste. I’ve walked through newer production lines that reclaim solvents, use less energy, and avoid dumping byproducts into rivers. Reworking the supply chain takes investment and patience, but public expectations keep that pressure front and center.
For people who care about creating smarter, safer products, the story behind a chemical like 1,1,1-Tris(4-hydroxyphenyl)ethane tells more than just what goes into plastic. It reflects choices made at every stage—from lab benches and factory floors to store shelves. Whether it shows up in a phone case, a medical device, or parts built to withstand the next storm, engineers and chemists carry responsibility for every molecule they mix. Clear facts, honest oversight, and a willingness to update old habits matter to anyone who likes products that last without causing harm.
Anyone spending time in a chemical lab, whether part of a research team, a student, or a production worker, knows that no substance is too ordinary to be treated with respect. 1,1,1-Tris(4-hydroxyphenyl)ethane stands as one of those chemicals that regularly turns up in various polymer and resin applications, but easy familiarity doesn’t mean skipping careful handling.
Wearing gloves seems like a no-brainer, but complacency creeps in once a routine settles. Long sleeves, a buttoned-up lab coat, and safety goggles form the front line. Skin contact matters for phenolic compounds, and these might cause irritation or, after repeated exposure, something longer-term. Once, back in my early days, I thought a quick transfer didn’t need face protection. I learned fast after a light splash led to minor but lasting irritation.
Gloves with strong resistance to chemicals—like nitrile—do a far better job than bare skin or casual latex. And chemical splash goggles have saved more than one eye from mishap. We lose nothing by being careful, but much by treating these steps as optional.
Nobody wants to spend the day breathing in powder or dust, especially when it can end up irritating airways or triggering allergies. Good ventilation in the lab, right down to using enclosed hoods and exhaust fans, pulls any airborne compounds away from breathing zones. Some labs monitor air quality and particulate levels after a few incidents of sneezing fits among staff. Even with something as seemingly straightforward as 1,1,1-Tris(4-hydroxyphenyl)ethane, keeping the air clean pays off in daily comfort and long-term health.
Once a spill happens and gets left unchecked, trouble follows. Clean work areas, clear walkways, and closed containers make any chemical, including this one, safer to handle. Storing the material in dry, labeled containers in a cool environment avoids accidental mixing, moisture pickup, or unnecessary exposure. It takes only a moment for a forgotten open jar to become a source for contamination or an unintended reaction. On a hot afternoon, I’ve seen powders shift and clump when left next to drying ovens. That small mishap made cleanup much tougher and wasted a good portion of an otherwise stable material.
Drop something, and the reaction time counts. Scrambling for paper towels gets no one far, and using vacuum cleaners without proper HEPA filters just blows dust into the air. Trained staff, equipped with spill kits stocked with absorbents and protective gear, tackle problems head-on and then manage clean-up waste as hazardous material, not common trash. Without a clear disposal process, these compounds end up lingering in places they don’t belong.
Cutting corners with chemical safety isn’t just about the rare accident; it’s about setting a habit worth repeating even on the least eventful day. Each of these precautions—gloves, goggles, airflow, secure storage—costs little compared to the headache of exposure or contamination. There’s no substitute for experience, but even well-trained hands gain from reminders: clean up after yourself, respect storage rules, and look out for the health of colleagues.
Curiosity moves science forward, and molecules like 1,1,1-Tris(4-hydroxyphenyl)ethane, often abbreviated as THPE, keep popping up in research papers and patent applications because of their unique properties. I remember first reading about THPE during my time in a university synthesis lab, marveling at its sturdy core structure and three extended arms capped with phenolic groups. This triphenolic compound isn’t just a page in a chemistry textbook—it makes a real difference in how industrial chemists build materials for everyday products.
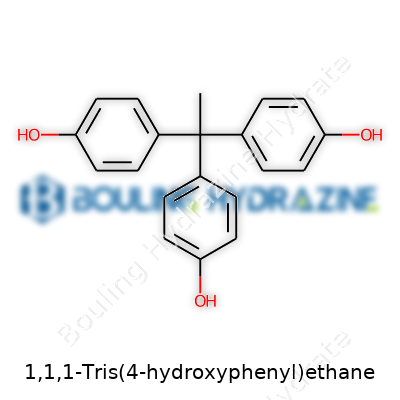
The molecular formula of 1,1,1-Tris(4-hydroxyphenyl)ethane is C20H18O3. Think of it as a central ethane backbone, where the central carbon hooks up with three phenyl rings at the 1-position of the ethane. Each of those rings carries a hydroxyl group in the 4-position, earning the “hydroxyphenyl” tag. Chemists sketch this structure almost automatically, picturing a three-arm shape with each arm hosting a reactive -OH group.
The layout gives THPE its particular functionality, making it a solid choice as a raw material in the production of high-performance resins and polymers. These polymers end up in windshields, electronics, adhesives, and specialty coatings. I’ve seen manufacturers depend on its rigid framework to deliver thermal stability and chemical resistance far beyond what basic plastic feedstocks can provide. Every time you swipe a scratch-resistant phone screen or open a laptop that stays cool at its edges, research like that around THPE plays a part.
The presence of three para-hydroxyl groups builds in strong antioxidant behavior. That means THPE traps free radicals and keeps chemical chains from breaking down under stress. In industrial curing processes, these functional groups help shape complex polymer networks. My professor once demonstrated a real-world use by comparing a batch of THPE-cured epoxy resins to an unmodified batch riddled with microfractures after a week at 120°C.
Every hydroxyl group reacts predictably with formaldehyde and other crosslinkers, giving materials scientists control over hardness, flexibility, and resistance to solvents. This keeps paint chips from flaking in sun-baked parking lots, or electronic boards from warping after a surprise coffee spill. And since THPE doesn’t introduce extra atoms into the system that degrade at low heat, it stretches out the usable lifespan of anything it touches.
The surge in demand for sustainable, high-performance plastics raises tough questions about safety and environmental impact. Phenolic compounds, including THPE, must meet strict handling standards due to their reactivity. I’ve joined colleagues in tackling these challenges, researching greener synthesis methods that minimize byproducts and reduce waste. There’s real progress in using catalytic systems that cut energy costs and shrink a lab’s footprint.
Better monitoring and improved purification also help keep contaminants out of the end products, making them safer for both workers and end-users. Responsible sourcing and handling, plus an honest look at the full lifecycle of THPE-derived products, make the difference between short-sighted profit and truly sustainable industrial chemistry.
I spent my early years in a university research lab, where every bottle on the shelf had written and unwritten rules. Some substances call for a little more attention. 1,1,1-Tris(4-hydroxyphenyl)ethane falls into that group. It’s neither the most hazardous, nor the easiest to ignore. With its role in polymers, adhesives, and specialty plastics, it’s a staple in advanced chemistry. But tossing it onto any old shelf isn’t safe or responsible.
Ask any lab worker who’s had to toss out a ruined batch: heat, light, and moisture will erode this compound’s reliability. Changes in color or clumping often signal a spoiled supply. Some compounds shake these things off, but not this one. Maintaining a dry, cool storage area keeps degradation away.
A platform away from windows and steam pipes makes a huge difference. On a forgotten shelf, UV rays may break molecular bonds. At best, this means wasting money on replacements. At worst, altered materials invite reaction mishaps. Refrigeration isn’t always necessary, but nobody regrets investing in temperature control, especially in summer.
Experience teaches that not all containers seal equally well. I’ve found wide-mouthed glass jars with strong lids work best to fend off moisture — plastic lets in more than people realize over time. Thick, amber glass does a good job blocking stray light. Materials that leach even a trace of plasticizer can react with hydroxyphenyl rings. Every time storage fails, the blame usually traces back to the wrong jar or a loose seal.
Mislabeling fuels accidents and unnecessary losses. Clear names and hazard pictograms make a world of difference when it matters most. Date every bottle when opening or receiving; this helps track changes over time. If a substance develops odd spots, saves time in checking age versus quality.
Labs often rotate drawers and move chemicals around, especially when busy. Consistent labeling reduces risk wherever life takes that jar. An unmarked or barely legible bottle in a back corner doesn’t help anyone.
Not everyone gets climate-controlled storage, especially in startups or educational labs. Dry cabinets or desiccators create adequate barriers if full-scale systems seem unattainable. Even a simple silica gel packet goes a long way for powders like this. For institutions with buying power, investing in trackable storage cabinets limits user error and theft, while building audit trails for compliance.
Managing ventilation reduces risks of dust, though this compound isn’t volatile. Localized extraction near weighing stations keeps the air safer, since no workplace should rely on luck or the hope that nothing spills.
Chemists, educators, and warehouse workers share a need: keep stock pure and available. Sloppy storage tells clients, regulators, and coworkers that corners get cut. Treating 1,1,1-Tris(4-hydroxyphenyl)ethane with care reassures that longevity, safety, and quality aren’t afterthoughts – they’re habits. In every lab I worked, these habits meant fewer mistakes and a lot less waste. It sounds simple, but it sets the best organizations apart.
Plenty of folks have never heard of 1,1,1-Tris(4-hydroxyphenyl)ethane. It pops up mostly behind the scenes, woven into the formulas for polymers and certain plastics that surround us every day. You might not see its name printed on a product label, but it can add strength and heat resistance to materials in electronics, construction, and consumer goods.
Conversations about chemicals like this often get a lot of attention, especially given growing anxiety about what’s in our food containers, water supplies, and even electronics. There’s a reason for ongoing public concern about chemical safety: the track record of industrial pollution and underreported risks isn’t just a relic of the past. Making sense of what’s dangerous and what’s not takes steady attention to facts, not just headlines.
Digging into the available research on 1,1,1-Tris(4-hydroxyphenyl)ethane, you run into a common problem: not much clear, public information on direct health impacts exists in peer-reviewed literature. The safety data sheets from chemical manufacturers often focus on prudent handling—wear gloves, use goggles, avoid dust inhalation. This advice fits most industrial powders and resins, not just this chemical. So far, there’s no widespread evidence linking this specific compound to cancer, hormone disruption, or acute toxicity in the way we’ve seen with substances like bisphenol A or certain phthalates.
Still, the absence of evidence doesn’t guarantee safety. Some compounds slip under the radar for years before deeper testing exposes longer-term issues. Since it shares some chemical similarities with other phenolic compounds, which can affect the body’s hormone systems, it makes sense to keep an eye on independent testing results.
Much of the conversation about chemical hazards today centers on what happens after a product leaves the factory—how it breaks down, whether it builds up in rivers or soil, and if it harms wildlife. For 1,1,1-Tris(4-hydroxyphenyl)ethane, there isn’t robust public data documenting persistence, bioaccumulation, or toxicity in ecosystems. That puts concerned communities and researchers in a tough spot: without evidence, nobody can say with certainty just how risky—or benign—environmental exposure may be.
In my own work in environmental reporting, I’ve learned that waiting for a clear signal of harm before acting can let problems grow unchecked. Our history with PCBs, dioxins, and other persistent organic pollutants shows that slow action often leads to higher cleanup costs and avoidable health problems down the line.
We can draw a lesson here: responsible innovation means staying curious and cautious, not complacent. If a chemical serves a function but its long-term health or ecological effects stay murky, it’s worth supporting more independent studies. Policymakers and industry partners could do their part by funding broad-spectrum tests and sharing results openly. Regular folks, from engineers to end consumers, have a role too—pushing for transparency and safer alternatives, especially when the science lags behind commercial use.
Trust grows when companies lay their research cards on the table and agencies act before problems hit crisis point. Until we see clearer proof of either safety or harm with 1,1,1-Tris(4-hydroxyphenyl)ethane, the smart move is to treat it like any chemical with unknowns attached: respect the risks, keep asking questions, and press for answers from those who turn raw chemicals into tools for daily life.